Ophthalmologists at UC Davis Health used an experimental gene therapy last month to treat a patient with wet age-related macular degeneration, or wet AMD. It was the first time the UC Davis Eye Center had used gene therapy.
The treatment was part of a randomized, partially masked, controlled, phase 3 clinical study evaluating the efficacy and safety of an experimental therapy, ABBV-RGX-314, for wet AMD. UC Davis Health is one of 93 sites in the U.S. participating in the clinical trial.
This investigational treatment is not FDA approved, and the efficacy and safety have not been established.
Wet AMD affects approximately 2 million people in the United States, Europe and Japan. It is a leading cause of vision loss among older adults.
“The current treatments for wet AMD may be life-long, and injections can be as frequent as every month,” said Glenn Yiu, professor of ophthalmology at UC Davis Health and principal investigator for the new clinical trial. “If approved, a gene therapy solution has the potential to maintain vision while reducing the number of injections, by allowing the eye to continuously produce the medicine on its own,” Yiu said.
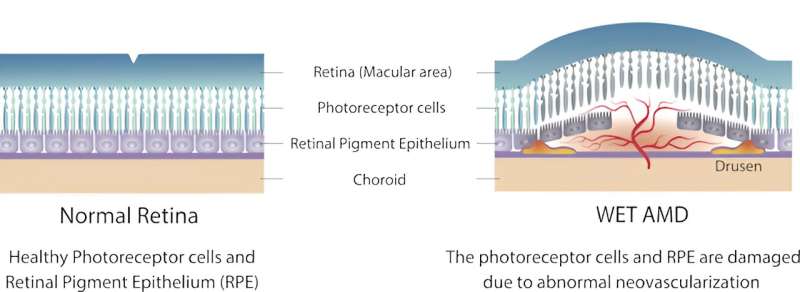
In AMD, the macula, an area of the eye’s lining that helps you see, becomes damaged. This can blur the central part of your vision, making it hard to drive or read. An early symptom of wet AMD is that straight lines look distorted and wavy.
In wet, or neovascular AMD, abnormal blood vessels grow underneath the retina. These vessels lead to bleeding or fluid leakage in the back of your eye, causing vision loss. This process, known as “neovascularization,” is largely driven by a growth factor called vascular endothelial growth factor (VEGF).
Treatments for wet AMD rely on repeated injections of drugs that block VEGF in the diseased eye.
Gene therapy may offer different approach
Unlike stem cell therapies used to treat eye diseases—which involve injecting cells with regenerative or restorative capabilities into the eye—gene therapy generally uses an empty viral envelope (a vector) to deliver a gene with specific genetic instructions for making protein.
ABBV-RGX-314 contains genetic instructions for making anti-VEGF proteins. After a single injection of ABBV-RGX-314 gene therapy, the eye can start to make the medicine on its own.
Yiu performed the first experimental gene therapy eye surgery at UC Davis Health in July. The procedure is more complex than administering a monthly injection. It includes a vitrectomy, where the viscous gel in the eye is removed and replaced with a saline infusion. The experimental treatment with its gene delivery vector is then injected underneath the retina.
Yiu will monitor whether the participant will continue to need monthly anti-VEGF injections in the coming months.
Paul Sieving is the former director of the National Eye Institute and is now a professor of ophthalmology at UC Davis Health. He established the Center for Ocular Regenerative Therapy (CORT) for pursuing cell and gene therapies.
“It is noteworthy for patients in Northern California that UC Davis Health is doing experimental ocular gene therapy studies in the Department of Ophthalmology and Vision Sciences. What excites me most about this is the potential of Dr. Yiu’s work to reduce the repeated eye injections currently required for wet age-related macular degeneration,” Sieving said.
UC Davis Health has enrolled three patients in the clinical trial and plans to enroll more. Individuals aged 50 to 88 with wet AMD who have had prior anti-VEGF injections may be eligible to participate.
Check out our AAV capsid engineering service to expedite your gene therapy research
PackGene is a CRO & CDMO technology company that specializes in packaging recombinant adeno-associated virus (rAAV) vectors. Since its establishment in 2014, PackGene has been a leader in the AAV vector CRO service field, providing tens of thousands of custom batches of AAV samples to customers in over 20 countries. PackGene offers a one-stop CMC solution for the early development, pre-clinical development, clinical trials, and drug approval of rAAV vector drugs for cell and gene therapy (CGT) companies that is fast, cost-effective, high-quality, and scalable. Additionally, the company provides compliant services for the GMP-scale production of AAVs and plasmids for pharmaceutical companies, utilizing five technology platforms, including the π-Alpha™ 293 cell AAV high-yield platform and the π-Omega™ plasmid high-yield platform. PackGene’s mission is to make gene therapy affordable and accelerate the launch of innovative gene drugs. The company aims to simplify the challenging aspects of gene therapy development and industrialization processes and provide stable, efficient, and economical rAAV Fast Services to accelerate gene and cell therapy development efforts from discovery phase to commercialization.
Related News
Exploring Tau Protein’s Role in Glaucoma: New Insights and Therapeutic Potential
Glaucoma, a chronic neurodegenerative disorder, leads to irreversible vision loss by damaging retinal ganglion cells (RGCs) and the optic nerve, often associated with increased intraocular pressure (IOP). Despite the benefits of IOP-lowering treatments, the underlying...
FDA-mandated CAR-T monitoring period could be halved, say researchers
In patients with diffuse large B-cell non-Hodgkin lymphoma (DLBCL), the two hallmark post-chimeric antigen receptor (CAR)-T therapy toxicities are extremely rare after two weeks, supporting a shorter, more flexible toxicity monitoring period, according to a study...
Ancestral CRISPR-Cas13 Ribonucleases Discovered: Implications for Genome Editing
In a pioneering study published in *Science*, a team of researchers led by Peter H. Yoon and Jennifer A. Doudna from the University of California, Berkeley, has made a remarkable discovery in the realm of CRISPR technology. The team has identified an ancestral clade...
KBI Biopharma Expands Manufacturing Contract with Global Pharmaceutical Company
KBI Biopharma Inc., a JSR Life Sciences company and global cGMP contract development and manufacturing organization (CDMO), has extended and expanded its manufacturing contract with a leading global pharmaceutical company. Originally initiated in 2020, the renewed...
Related Services

Plasmids GMP Services
Multiple scales & grade of solutions of various kind of plasmids suitable for multiple treatments in a fast and cost effective way.
READ MORE

AAV GMP Services
Ranging from small-scale AAV production, to large-scale AAV cGMP manufacturing for animal studies.
READ MORE

Technology Platforms
PackGene’s proprietary π-Alpha™ 293 AAV High-yield Platform increases AAV production by 3 to 8 times that of traditional platforms.
READ MORE

