Key Benefits
-
World-Class Quality
Our mRNA and LNP products meet top CDMO standards -
Proven Track Records
Trusted performance for seamless drug development -
Strict Quality Control
Comprehensive testing ensures purity and consistency
What We Offer
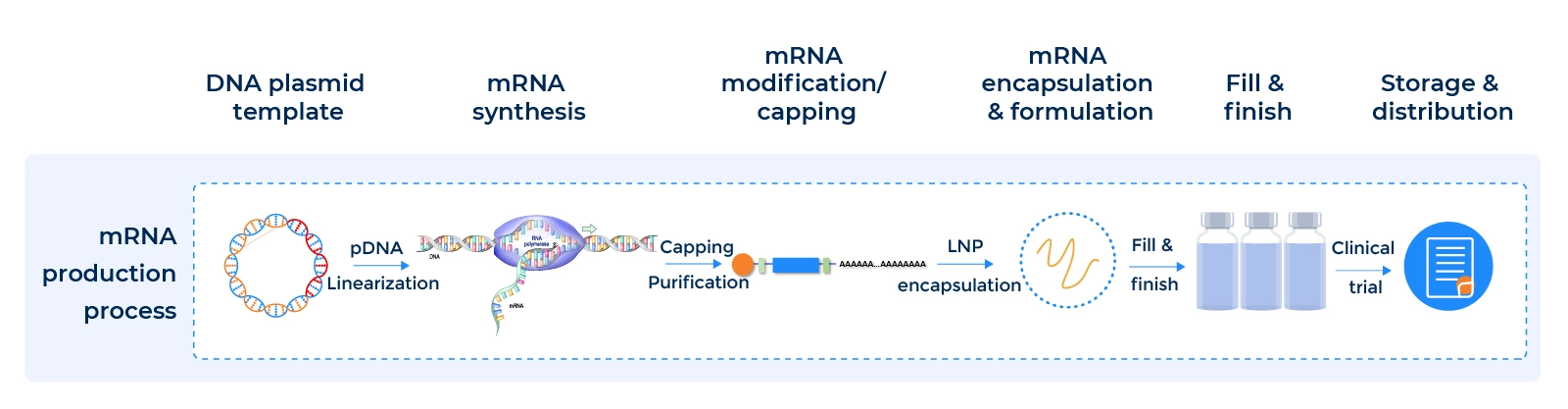
At PackGene and Kudo Biotechnology, we are committed to delivering the highest quality GMP mRNA production process. Our process starts with GMP plasmid DNA manufacturing, which includes the creation of an E. coli strain cell bank, fermentation, plasmid DNA purification, and QC release to ensure that the DNA is of the highest quality.
Next, we move on to mRNA transcription and purification, where we linearize the pDNA and perform in-vitro transcription, capping, dsRNA removal, bulk purification, and QC release to ensure that the mRNA is pure and free from contaminants.
For all your mRNA manufacturing needs. Our streamlined process ensures ease in your CMC
Performance
-
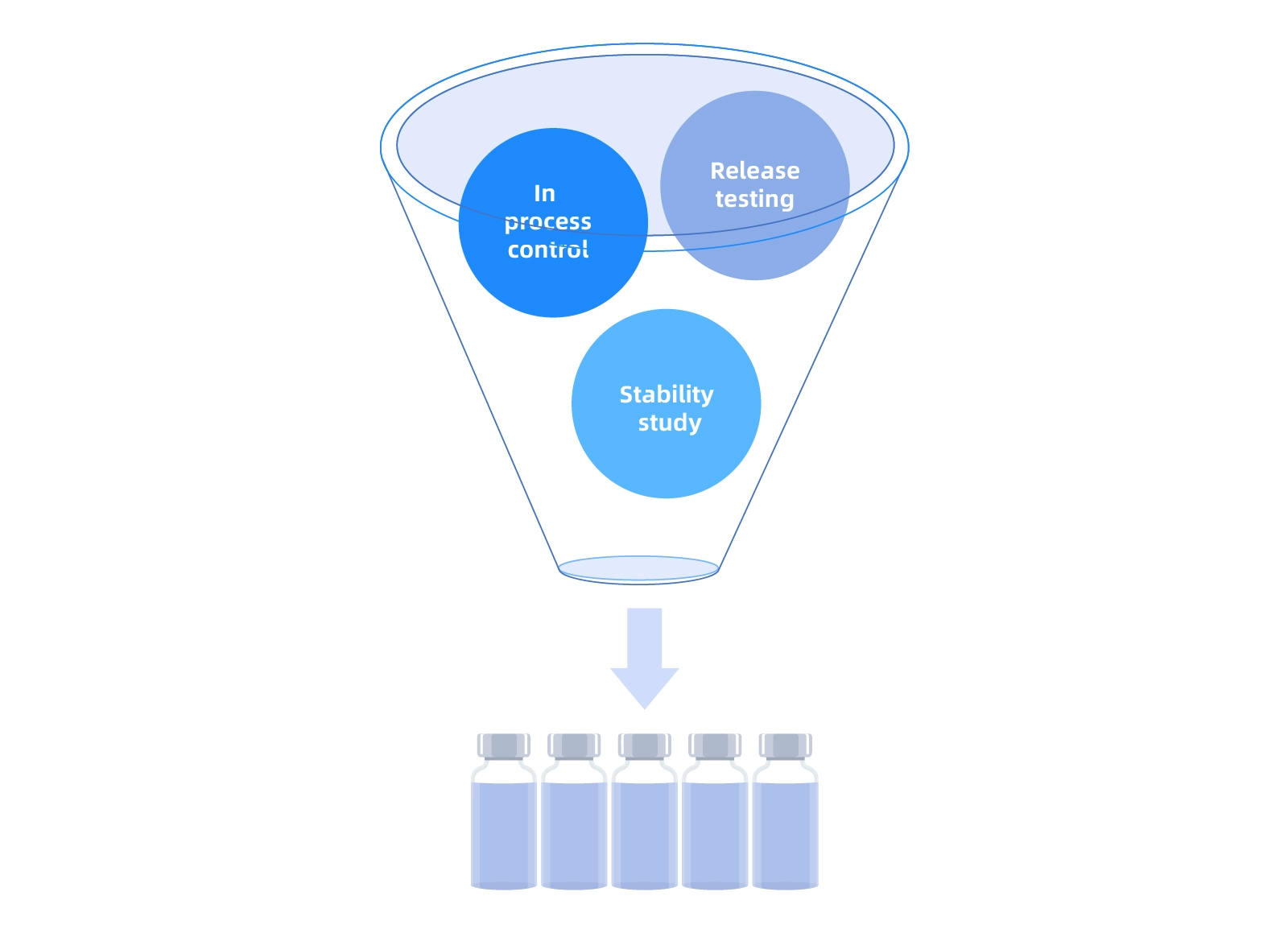
Product Quality and Speed
Performance

Speed
Quality

-
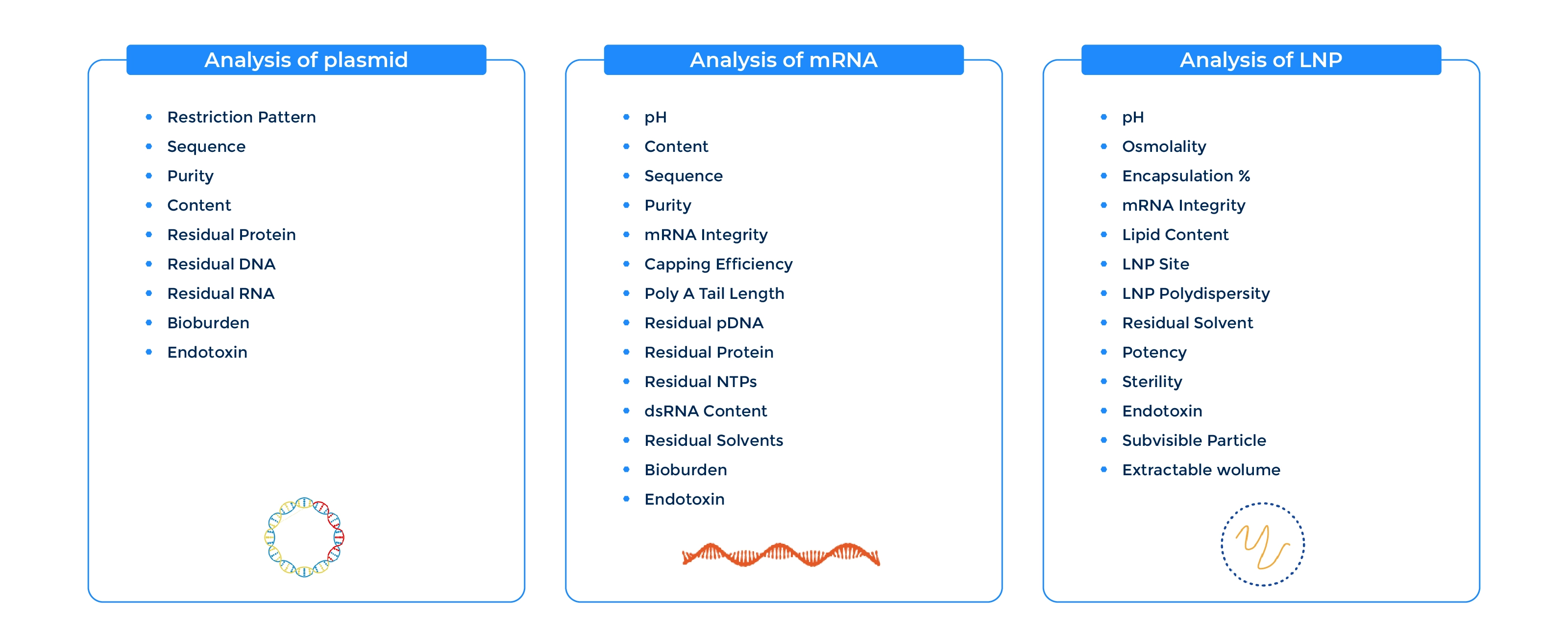
Comprehensive Analytical Panel
-
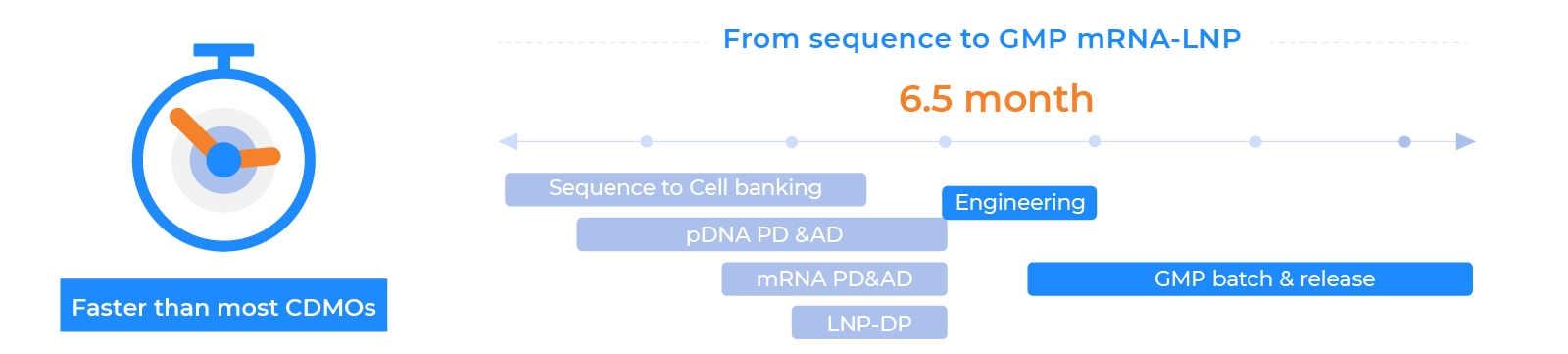
Project Timeline
-
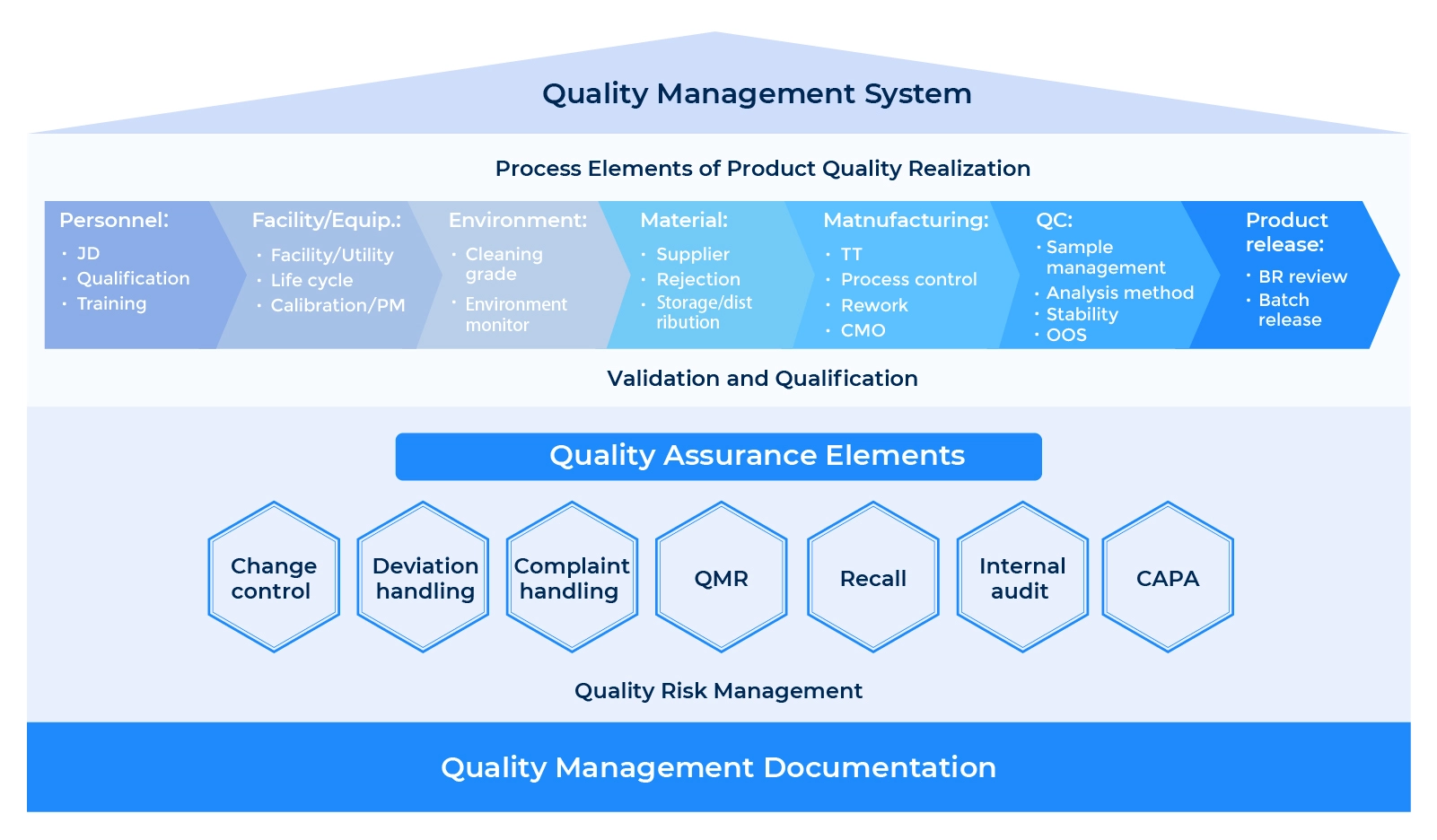
Quality Management System
-
Documentation
Facility
Plasmid cGMP Manufacturing Site
- 1,300 sqf PD room
- 11,000 sqf GMP room
- 5L to 200L fermenters
- Batch sizes of up to ~100g
PackGene’s state-of-the-art GMP plasmid production site is dedicated to providing reliable, compliant, and scalable plasmid production services. With a team of ~40 staff members, including management staff members with over 15 years of experience and over 7 years of AAV and plasmid prep experience. The site and staff ensure that reliable materials are generated for the production of high-quality mRNA, and our team takes shifts to ensure continuous production without interruption.
The facility is equipped with fermenters ranging from 5L to 200L, enabling batch sizes of up to ~100g plasmid, with hundreds of lots produced yearly.
PackGene s plasmid production site has a 1,300 sqf PD room and a 11,000 sqf GMP room, ensuring compliance with regulatory standards. With its reliability, scalability, and compliance, the facility provides high-quality plasmid production services that exceed industry standards.

Related Service
REQUEST A QUOTE
Confidentiality Commitment from PackGene:
The information you submit here will be kept strictly confidential. Packgene will not disclose to any third party or related personnel, and it will only be used for project evaluation and progress reports according to the requests from submitter under confidentiality conditions.