Key Benefits
-
Productivity
Two independent cGMP plasmid production lines support scalable plasmid DNA manufacturing up to 200L — ideal for both clinical trial supply and commercial programs. -
Quality
Isolated production suites, advanced cleaning procedures, and strict use of disposable materials ensure complete avoidance of cross-contamination, delivering consistently pure, regulatory-grade plasmids. -
Flexibility
Multiple plasmid production scales (pilot to large volume) and grades (research, NHP, GMP) allow tailored solutions to meet your project requirements efficiently and cost-effectively. -
Expertise
PackGene’s specialized plasmid manufacturing team brings over 10 years of experience in high-yield, clinical-grade plasmid DNA production, ensuring technical excellence and robust regulatory compliance. -
Advanced equipment
Industry-leading single-use technology is deployed in both upstream and downstream processing. All fill & finish operations are performed under VHP (Vaporized Hydrogen Peroxide) isolators for maximum safety and sterility.
Service Details
GMP Plasmid Manufacturing Facility
PackGene is a world-leading GMP plasmid manufacturer, with state-of-the-art facilities purpose-built for the highest standards of cGMP plasmid DNA production. Our 1,300 sq ft Process Development (PD) lab serves as the innovation hub for advanced R&D in plasmid process development, optimization, and scale-up.
Our impressive 11,000 sq ft GMP manufacturing suite is equipped with advanced fermenters ranging from 5L to 200L – enabling batch sizes, high-yield runs, and manufacturing of up to 100g per batch. With the capacity to produce more than 1,000g of GMP-grade plasmid DNA annually, PackGene is ready to meet the supply needs of the most ambitious clinical and commercial gene therapy, cell therapy, and vaccine projects.
Supporting this output is a dedicated, expert team of ~40 staff decades of combined experience in AAV plasmid preparation and GMP operations. We maintain seamless, uninterrupted production with 24/7 shift operations - ensuring the reliability, quality, and scalability our partners require.
PackGene’s commitment is to deliver top-quality plasmids —produced, tested, and released under strict regulatory compliance— to advance cell and gene therapy research, clinical development, and commercial innovation.

Performance
-
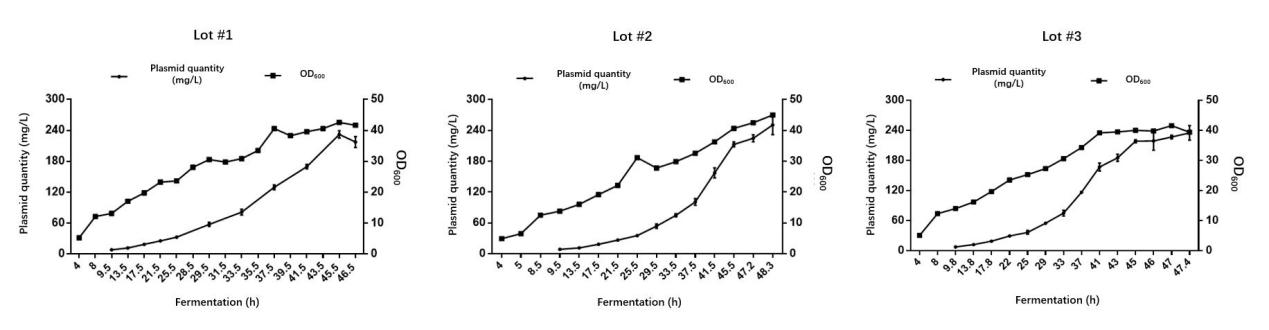
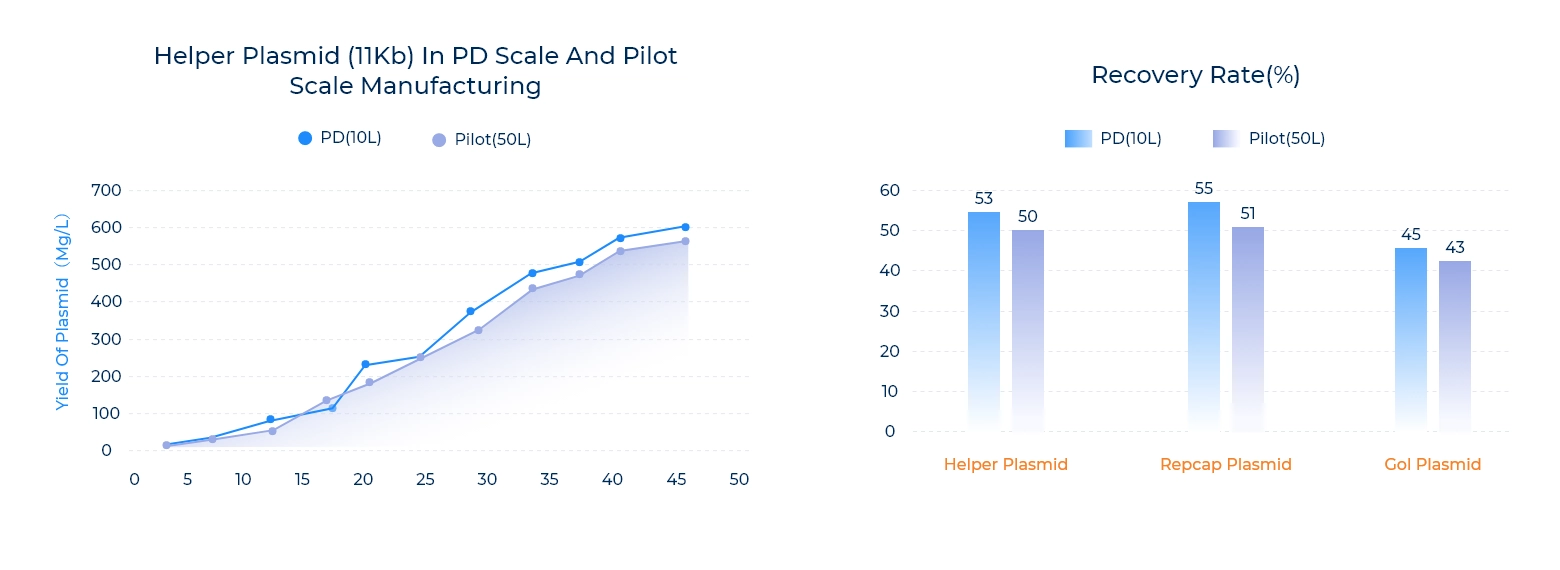
Scalability
PackGene’s ongoing optimization of GMP plasmid production processes ensures exceptional scalability—delivering consistent , high-yield batch production for every project. Our advanced fermentation and purification workflows support seamless scale-up from research to clinical and commercial lots, driving reliable batch-to-batch yield and recovery rates for GMP plasmid DNA manufacturing.
-
Batch-to-batch consistency
Related Service
REQUEST A QUOTE
Confidentiality Commitment from PackGene:
The information you submit here will be kept strictly confidential. Packgene will not disclose to any third party or related personnel, and it will only be used for project evaluation and progress reports according to the requests from submitter under confidentiality conditions.