Key Benefits
-
Endotoxin and Contamination control
-
Precisely quantified Titer, Genome copy and integrity
-
Lower safety risks and enhance efficacy
-
Reproducible results
Service Details
| AAV type | Scale | Turnaround time |
|---|---|---|
| Standard capsids (guaranteed yield) | 1E+13 GC~8E+15 GC | Start from 17 business days |
| Custom capsids (by volume) | Up to 100L with 30mL-200mL small scale test | Start from 25 business days |
AAV yield depends on factors like serotype and gene of interest. We offer guaranteed packages for high-yield serotypes (1, 3b, 5, 7, 8, 9, Rh10). For other serotypes, we produce based on volume and can conduct small-scale tests to determine necessary production. Contact technical support for more details.
QC Standards
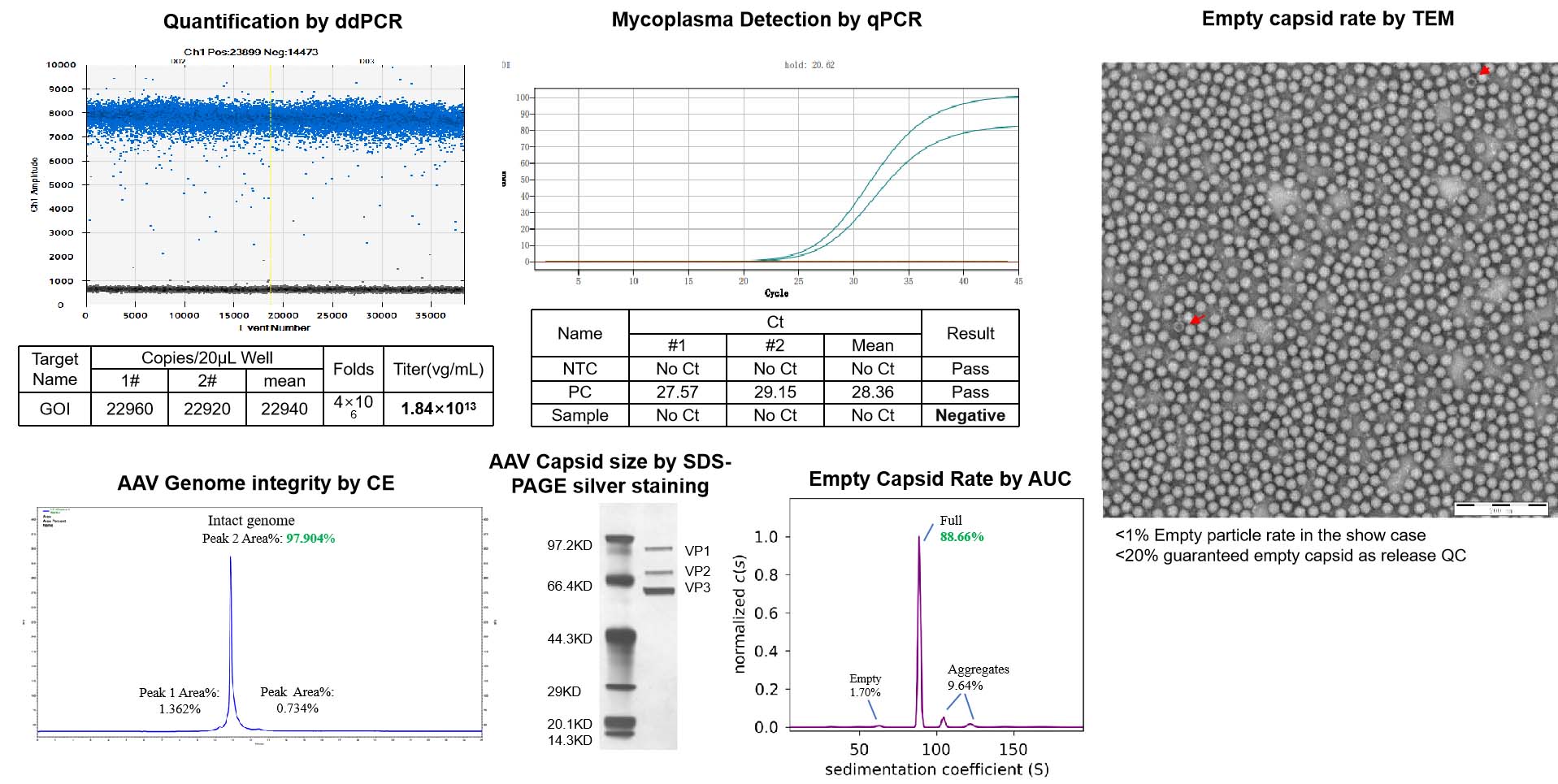
NHP grade AAV undergoes stringent QC tests before being released to ensure the quality of the AAV product and minimize the adverse effects in animal studies.
| Test | Method | QC Standard | |
|---|---|---|---|
| Standard QC | ddPCR | Measure titer, normalized to 1e13vg/ml | Standard capsids: Concentration and total quantity meet needs. Custom capsids: quantity based on production scale. |
| AAV Capsid size* | SDS-PAGE silver stain | Match capsid protein size | |
| Guarantee endotoxin | LAL | <1EU/ml | |
| Mycoplasma Detection | qPCR | Negative | |
| Bioburden | Direct inoculation | No growth | |
| AAV Genome integrity | CE(titer>1e+12vg/ml, volume >50ul) | Report | |
| Empty Capsid Rate | TEM | <20% | |
| Additional QC | AAV Genome sequencing | Nanopore | Report |
| Empty Capsid Rate | AUC | Report | |
| Endotoxin removal | LAL | <0.2EU/ml | |
| Residual Triton Analysis (bundle with endo removal) | HPLC | 5ppm | |
| Sterility test | Direct inoculation | No growth |
Performance
-
Comprehensive Testing for AAV Quality, Purity, and Safety, Ensuring Suitability for NHP Studies
-
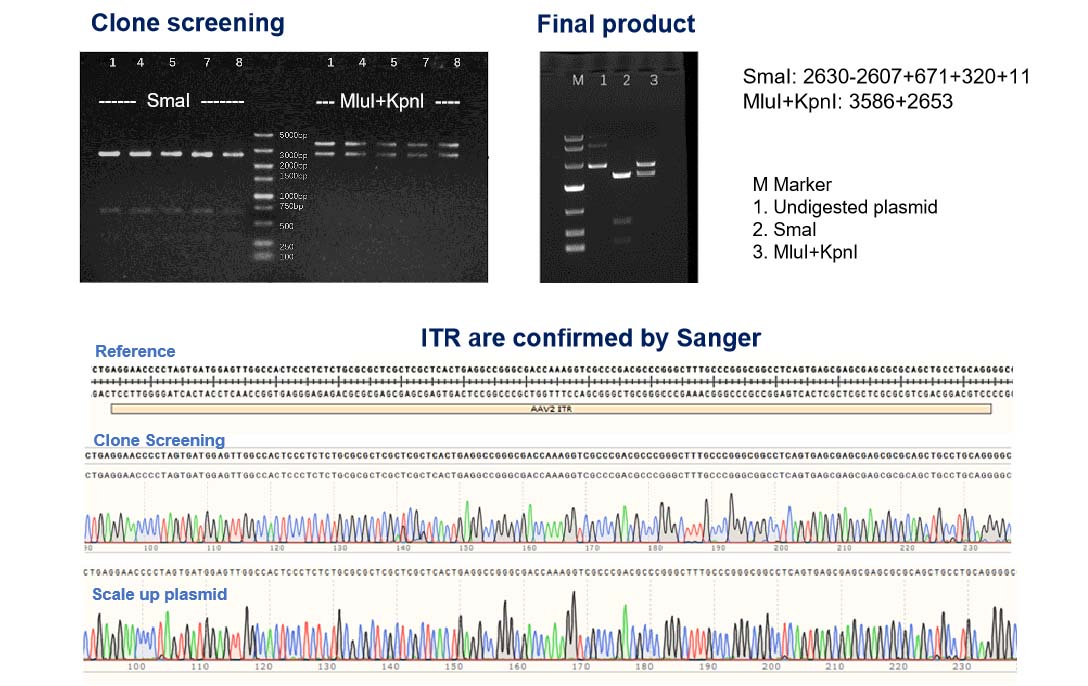
Ensuring Plasmid Integrity: Double-Check of ITR and GOI for High-Quality NHP-Grade AAV Production