Overview
About Adeno-Associated Virus (AAV) – Why It’s the Gold Standard for Gene Therapy Research
Adeno-associated virus (AAV) is a non-pathogenic, single-stranded DNA virus that has emerged as an exceptional research tool and leading candidate for genetic payload delivery in gene and cell therapy applications. Several features make AAV the preferred vector for both academic and industry researchers:
- Non-pathogenic Safety: AAV is not currently known to cause any disease in humans, making it an ideal choice for in vivo and in vitro gene delivery.
- Low Immunogenicity: AAV infection results in a very mild immune response, supporting higher dosing, long-term gene expression, and minimal off-target effects for research-grade and clinical-grade applications.
- Broad Tropism: Unique among viral vectors, AAV can efficiently infect both dividing and non-dividing cells—greatly expanding its utility across diverse tissue and cell types.
- Stable, Durable Expression: Recombinant AAV vectors drive prolonged gene expression without integrating the gene of interest (GOI) into the host genome, minimizing safety concerns and enabling sustained therapeutic effect.
- Serotype-Driven Specificity: Differences in the capsid structure of various AAV serotypes bias infection rates across host cell types, offering precise tissue or cell-type targeting for gene therapy and research use.
Key Benefits
-
Fast Turnaround
Miniprep to virus in as little as 12 days—maximize project velocity with industry-leading viral vector production timelines. Perfect for biotech startups, academic labs, or pharmaceutical pipeline acceleration. -
High Purity
Each batch undergoes rigorous SDS-PAGE verification—guaranteeing contaminant-free AAV vector prep for reliable in vivo and in vitro gene delivery. -
Low Endotoxin
<10 EU/mL—GMP-ready and safe for animal studies, supporting IND-enabling, preclinical, and translational R&D projects. -
Low Empty Capsids
Consistently <30% empty shells—superior genome integrity and transduction efficiency in cell culture, mouse, or rat gene therapy models. -
Serotype Variety
Choose from 70+ custom AAV serotype options—enabling precision targeting for brain, liver, muscle, retina, or systemic applications. Supports both tissue-specific and broad tropism projects. -
Expert Support
Partner with PhD-level viral vector specialists—guidance across construct design, optimization, troubleshooting, and regulatory consulting for over 50,000 successful customer projects worldwide. -
Best Viral Vector CRO/CDMO Partnership
Leverage PackGene’s reputation as a top-rated CRO for custom vector production, scalability to GMP manufacturing, and advanced analytical QC.
Service Details
Price and Turnaround
The indicated titers are guaranteed* except when the insert exceeds the packaging capacity (4.7 kb) or if you choose to provide us with your own modified rep/cap plasmid or helper plasmid.
| AAV Packaging Serotypes | Guaranteed Yield (GC)* | Lead Time (Business Days) |
|---|---|---|
| Normal-yield AAV Serotypes | 2E+12 GC | 12-15 Days |
| 5E+12 GC | ||
| 1E+13 GC | ||
| 2E+13 GC | ||
| 5E+13 GC | ||
| 1E+14 GC | ||
| 2E+14 GC | 18-24 Days | |
| 5E+14 GC | ||
| 1E+15 GC | 30-45 Days | |
| 2E+15 GC | ||
| Low-yield AAV Serotypes (AAV4, 6, etc.) | 2E+12 GC | 12-15 Days |
| 5E+12 GC | ||
| 1E+13 GC | ||
| 2E+13 GC | ||
| 5E+13 GC | ||
| 1E+14 GC | 18-24 Days | |
| 2E+14 GC | ||
| 4E+14 GC |
- GC = Genome copies.
- For these extremely low-yield AAV serotypes without production data, we are not able to guarantee the final yield or titer specified here.
- If you would like to use any AAV serotypes that are currently under patent, and your application is for commercial use, we advise that you contact the patent owner to obtain authorization beforehand.
- *For our guaranteed yield AAV packaging, if the project yields are confirmed to be significantly below expectation after two production attempts, we will proceed to close the project and deliver the available material. We appreciate your understanding and cooperation.
Storage Requirements:
- Store the virus at -80°C, and place it on ice during operation.
- Calculate your expected usage in advance and PackGene will aliquot your virus according to your pre-determined requirements. This can help avoid unnecessary thawing and re-freezing after receiving your AAVs since freeze thaw cycles influence virus viability. If aliquoting is required, it is recommended to use PCR tubes with siliconized inner walls, or special virus preservation tubes with low protein binding rates.
- Thaw your virus aliquots in an ice bath immediately before use.
- Dilute with PBS or PBS / 0.001% F-68 if needed.
Quality Control
PackGene’s experienced QC team has developed a comprehensive suite of AAV-specific quality control assays to verify the identity, purity, and potency of AAV viral particles for both in vitro and in vivo studies. Our advanced QC panel supports rigorous research, preclinical, and GMP manufacturing requirements—helping ensure the safety and performance of your gene therapy vectors.
- AAV Genome Quantification: Genome copies are precisely measured using SYBR qPCR, calibrated against ATCC’s Reference AAV, providing accurate titer assessment.
- Purity Analysis: Capsid protein purity is assessed via Coomassie-Blue staining, confirming the integrity and quality of the viral preparation.
- Endotoxin Guarantee: All AAV batches are delivered with guaranteed endotoxin levels below 10 EU/mL, making them suitable for sensitive animal and clinical studies.
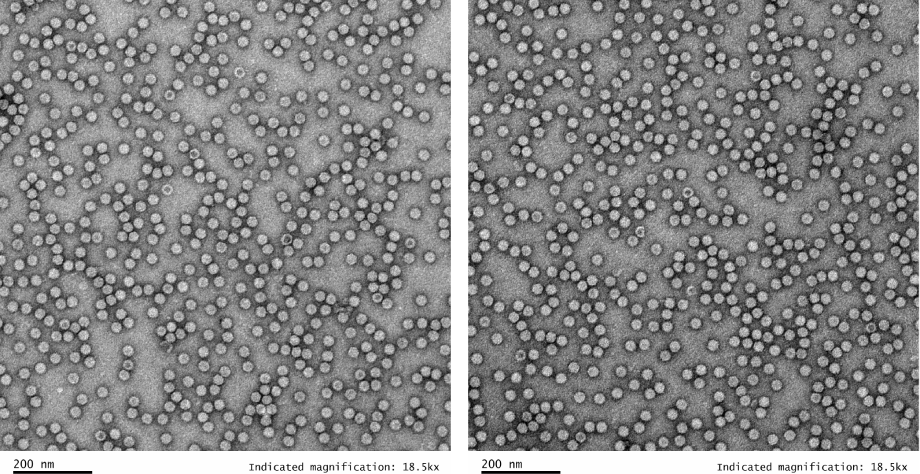
- Additional QC Services: Expanded testing options include ddPCR for high-resolution quantification, TEM imaging for pa rticle morphology, TCID50 for infectivity titering, and a robust menu of analytical QC services.
- Explore our full AAV Analytical Services for more details on PackGene’s validated QC assays and custom testing capabilities.
Our QC infrastructure enables high-confidence lot release and supports IND/BLA submissions, regulatory compliance, and reliable gene and cell therapy development
| Category | QC Assays | QC Standard |
|---|---|---|
| Identity | Identity – GOI Sequence | Additional QC |
| Purity | SDS-PAGE Coomassie Blue Staining | Free QC |
| TEM | Additional QC | |
| AUC | Additional QC | |
| Potency & Content | qPCR | Free QC |
| ddPCR | Additional QC | |
| TCID50 | Additional QC | |
| Capsid Titer-ELISA | Additional QC | |
| Impurity | Endotoxin Test | Free QC |
| Mycoplasma Detection | Additional QC | |
| Sterility Test | Additional QC | |
| Residual Plasmid Test | Additional QC |
Performance
-
Standard QC
- Restriction digestion analysis using multiple endonuclease to verify the plasmids to be used for AAV packaging.
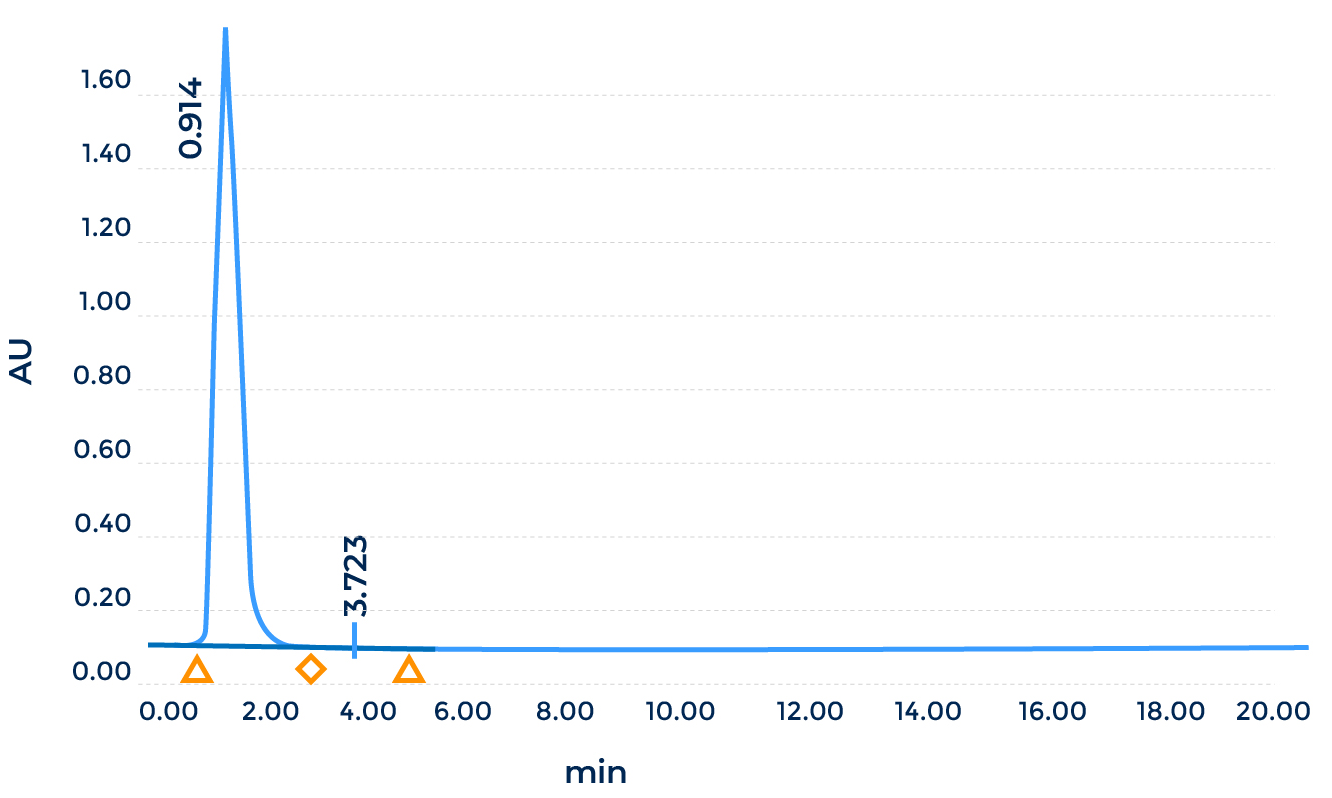
- AAV Titering by qPCR (SYBR Green with standard curve for quantification)
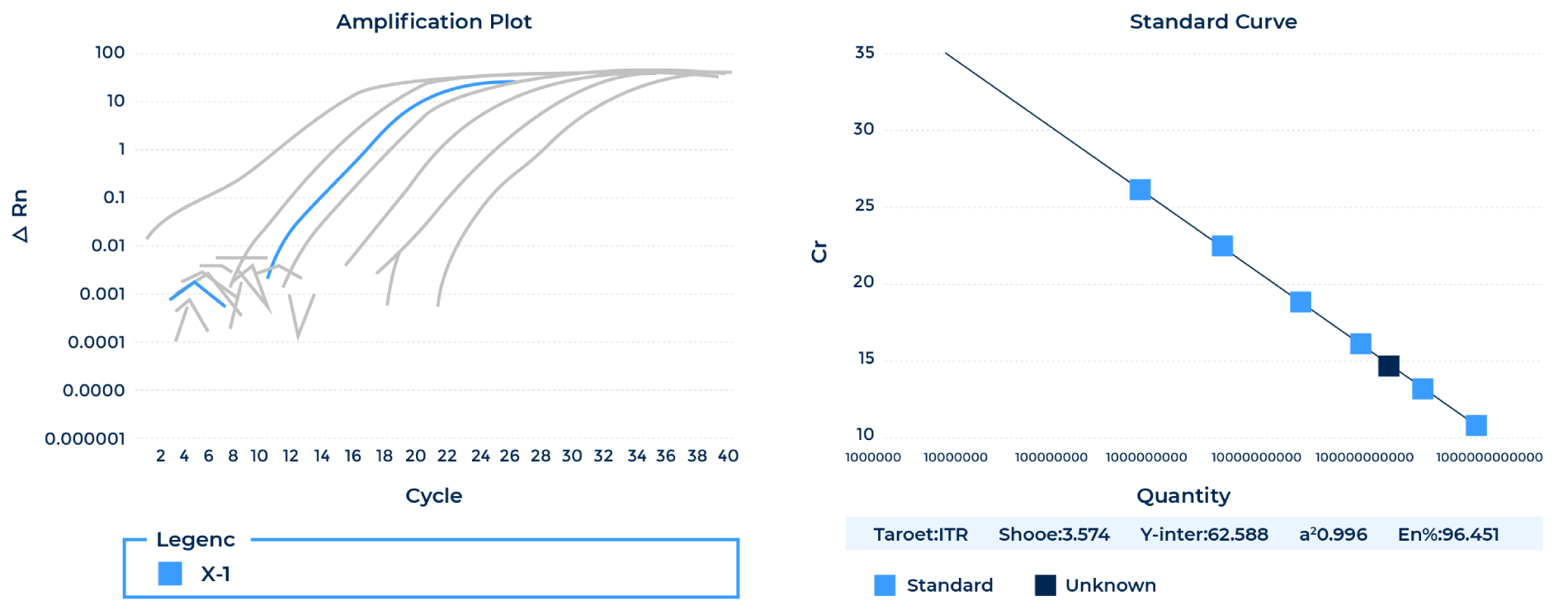
Note: ATCC VR-1816™ was used as the standards for AAV qPCR titering.- Endotoxin Test by LAL assay
- AAV purity analysis by SDS-PAGE and Coomassie Staining (silver staining available upon request)
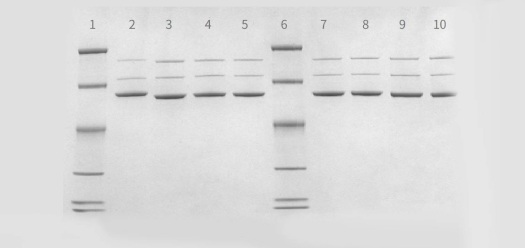
Legend: Lanes 2-5 and 7-1: AAV samples produced at PackGene. Lane 1,6: Marker -
Other Available Analytical Tests