Key Advantages
-
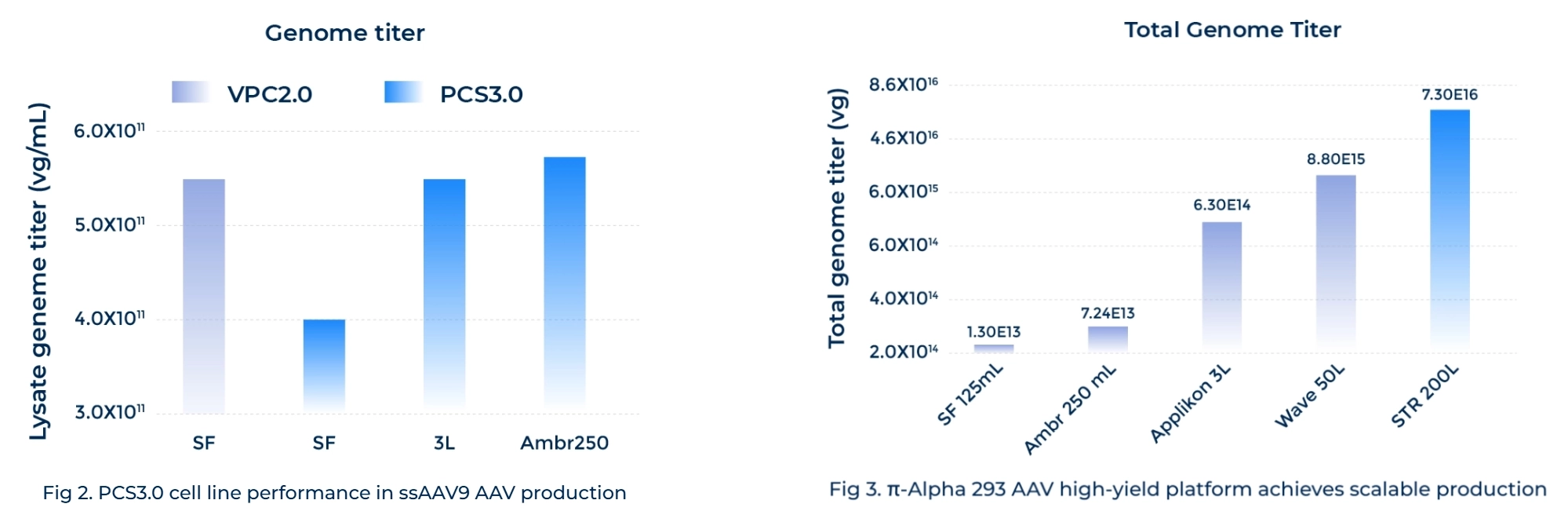
Enhanced Productivity and Scalability
Proprietary technologies drive a greater than 10-fold increase in AAV yield within HEK293 serum-free cell suspension systems. -
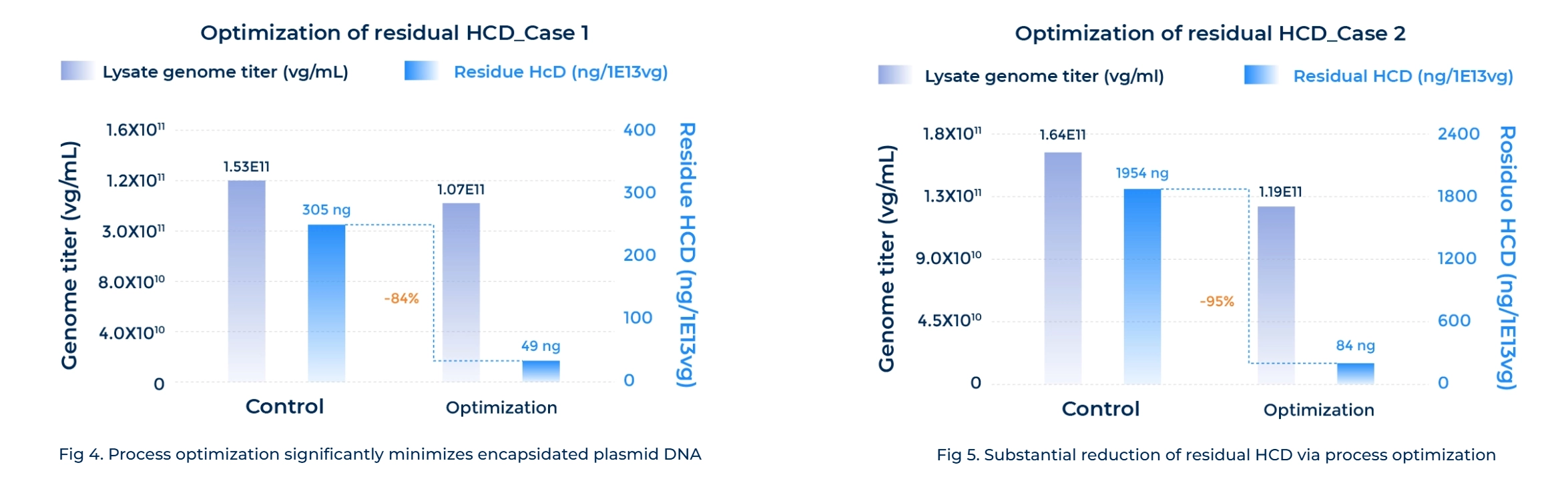
Superior Purity
Refined process development significantly reduces key impurities, including Host Cell DNA (HCD) and endotoxin. -
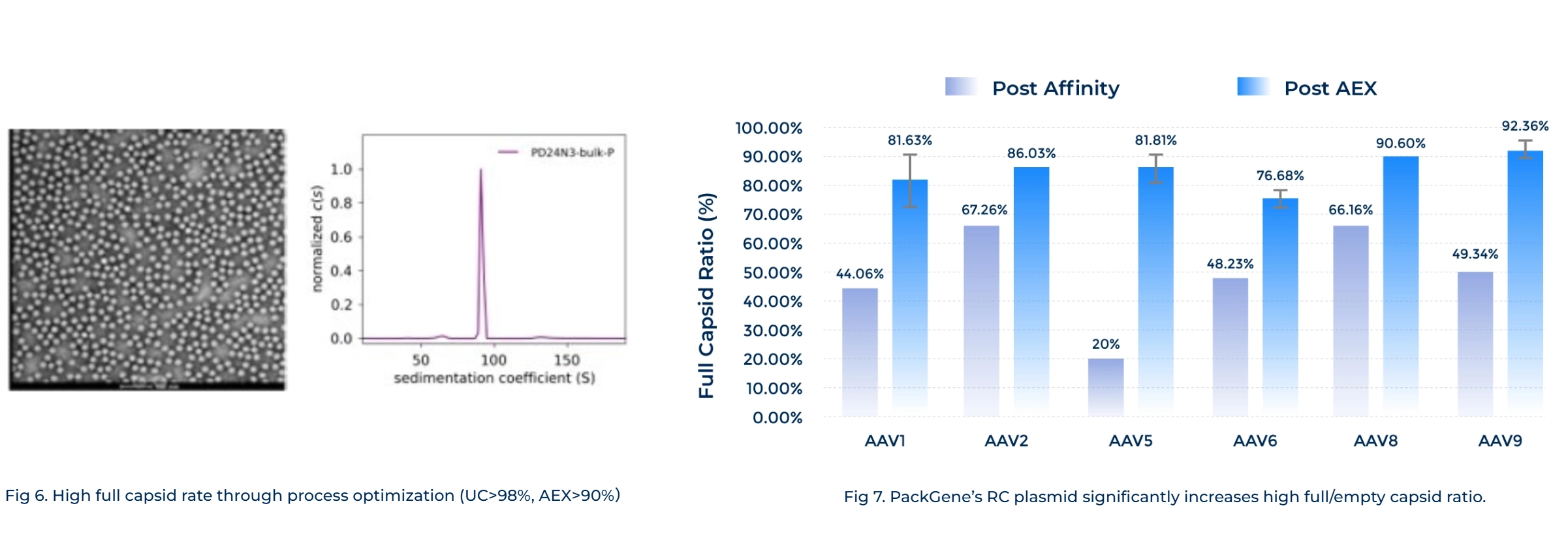
Improved Safety and Bioactivity
Innovative technological advancements minimize empty capsid and maximize infection titers.
Performance
-
Enhanced Productivity and Scalability
The PCS3.0 suspension cell line, combined with a uniquely engineered RC plasmid, enhances AAV production efficiency. This optimized system enables greater scalability for large-scale manufacturing.
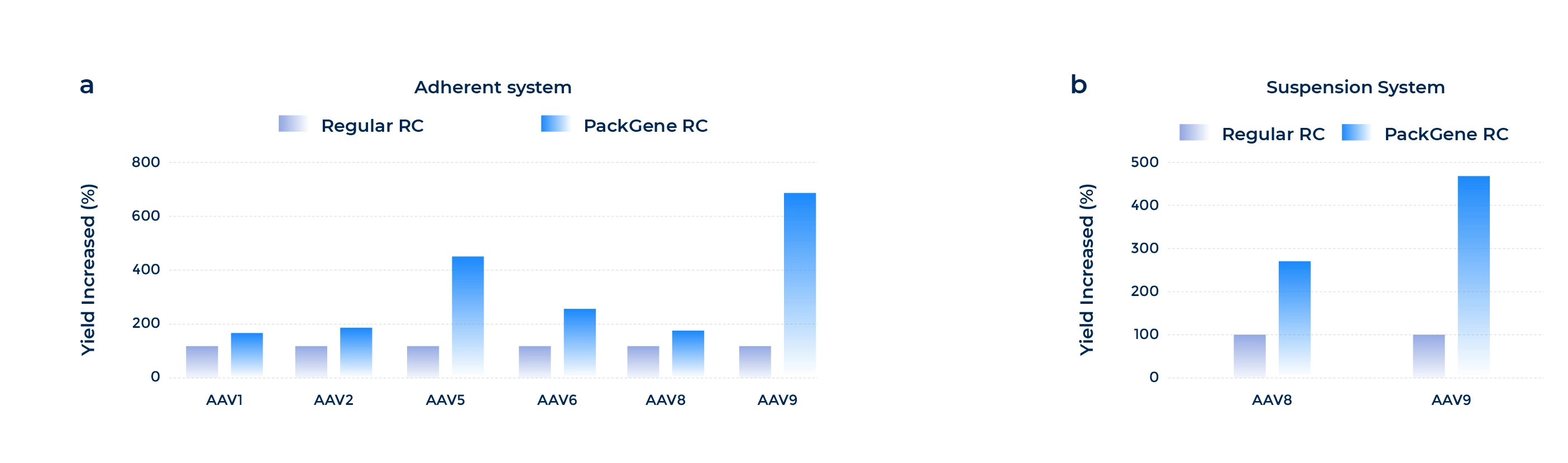
Fig 1. PackGene’s proprietary RC plasmid significantly increases AAV production yield
-
Superior Purity
-
Improved safety and bioactivity