Gene therapy that induces the body to create microRNA-22 (miR-22), a naturally occurring molecule, successfully treated mice with hepatocellular carcinoma, the most common form of liver cancer.
The miR-22 treatment also reduced liver inflammation and produced better survival outcomes with no observable toxicity compared to the FDA-approved liver cancer treatment lenvatinib.
Those are the findings of a new study from the UC Davis Comprehensive Cancer Center published in Molecular Therapy.
“This research introduces miR-22 gene therapy as a promising and innovative approach for treating hepatocellular carcinoma,” said Yu-Jui Yvonne Wan, senior author of the study. Wan is a distinguished professor and vice chair for research in the UC Davis Department of Pathology and Laboratory Medicine. “The study’s findings suggest that miR-22 therapy could provide better survival outcomes, enhance anti-tumor immunity, improve metabolism and reduce inflammation.”
The University of California filed a patent application for Wan’s discovery of miR-22 for treating hepatic and metabolic diseases. The patent is currently pending.
MicroRNAs’ role in health and disease
MicroRNAs are small molecules that contain ribonucleic acid (RNA), a type of genetic material. MicroRNAs are widely found in plants and animals. They are “non-coding,” meaning they do not make proteins like some other RNA molecules.
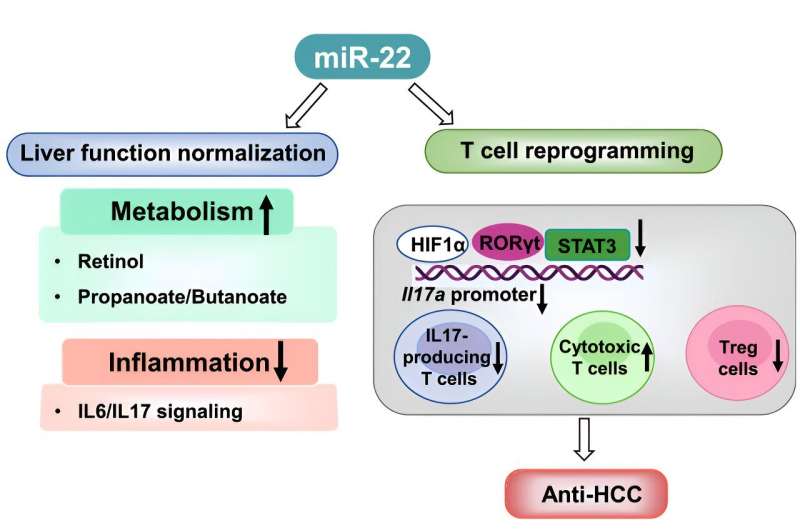
In the case of miR-22, it acts like a brake, stopping the production of certain proteins, including cyclin A, protein deacetylases and hypoxia-inducible factor, that can fuel liver cancer growth. A lack of miR-22 is found in hepatocellular carcinoma tumors, and its levels can predict survival time for patients with the disease.
Wan studies the interaction between gut microbes and the liver, what’s known as the gut-liver axis. Her previous research shows that metabolites produced by the gut microbiome help create miR-22 and have anti-cancer benefits. These metabolites include retinoic acid, short-chain fatty acids, bile acids and vitamin D3.
When people have colon or liver cancer, signaling from these metabolites is reduced, which in turn reduces miR-22.
This was a clue for Wan that returning miR-22 to a higher level might act as a treatment for liver cancer. She launched several studies, including one demonstrating miR-22 could suppress liver tumors by targeting both liver and immune cells.
Gene therapy to test efficacy of miR-22 for liver cancer
For this study, Wan and first author Ying Hu, an assistant professional researcher in Wan’s lab, turned to gene therapy.
Using an inactivated adenovirus, they introduced miR-22 into the mice with a single intravenous injection.
The mice treated with the gene therapy were compared with mice treated with the current FDA-approved drug lenvatinib (administered orally once a day), untreated mice and healthy mice.
Both miR-22 and lenvatinib inhibited the progression of the liver cancer compared to untreated mice. However, the miR-22-treated mice had longer survival times without toxicity compared with lenvatinib-treated mice. Some of the study’s key findings:
- MiR-22 and lenvatinib reduced serum alanine transaminase (ALT), aspartate aminotransferase (AST), and cholesterol levels, indicating improved liver function.
- MiR-22-treated mice showed no blood or organ toxicity.
- At five weeks, untreated mice had enlarged livers, making up 33.5% of their body weight. The treated mice had smaller, less diseased livers, with ratios of 10.9% (miR-22) and 12.0% (lenvatinib) of body weight.
- The median survival rate was 46 days for the lenvatinib group and 50 days for the miR-22 group.
- Two mice treated with miR-22 remained alive for 60 days. In contrast, the survival time for the untreated mice was about 40 days.
Liver cancer is a leading cause of death worldwide
According to the American Cancer Society, liver cancer is a leading cause of cancer deaths worldwide, accounting for more than 700,000 deaths each year. In the United States, each year about 25,000 men and 11,000 women are diagnosed with liver cancer, while 19,000 men and 9,000 women die from the disease.
Risk factors for liver cancer include long-term hepatitis B or hepatitis C infections, obesity, non-alcoholic fatty liver disease, excess alcohol consumption, auto-immune liver disease and exposure to aflatoxins (a toxin found in moldy peanuts or grains).
Hepatocellular carcinoma is becoming a bigger problem due to rising obesity rates worldwide, including in the U.S.
Drugs to treat hepatocellular carcinoma—sorafenib, lenvatinib, regorafenib, and cabozantinib—are associated with considerable toxicities and poor quality-of-life outcomes. The survival benefit is limited to a few months and the cost is very high.
“The positive findings from this preclinical study give us hope that miR-22 could be a promising alternative to treat hepatocellular carcinoma,” Hu said.

Check out our AAV CDMO service to expedite your gene therapy research
PackGene is a CRO & CDMO technology company that specializes in packaging recombinant adeno-associated virus (rAAV) vectors. Since its establishment in 2014, PackGene has been a leader in the AAV vector CRO service field, providing tens of thousands of custom batches of AAV samples to customers in over 20 countries. PackGene offers a one-stop CMC solution for the early development, pre-clinical development, clinical trials, and drug approval of rAAV vector drugs for cell and gene therapy (CGT) companies that is fast, cost-effective, high-quality, and scalable. Additionally, the company provides compliant services for the GMP-scale production of AAVs and plasmids for pharmaceutical companies, utilizing five technology platforms, including the π-Alpha™ 293 cell AAV high-yield platform and the π-Omega™ plasmid high-yield platform. PackGene’s mission is to make gene therapy affordable and accelerate the launch of innovative gene drugs. The company aims to simplify the challenging aspects of gene therapy development and industrialization processes and provide stable, efficient, and economical rAAV Fast Services to accelerate gene and cell therapy development efforts from discovery phase to commercialization.
Related News
Exploring Tau Protein’s Role in Glaucoma: New Insights and Therapeutic Potential
Glaucoma, a chronic neurodegenerative disorder, leads to irreversible vision loss by damaging retinal ganglion cells (RGCs) and the optic nerve, often associated with increased intraocular pressure (IOP). Despite the benefits of IOP-lowering treatments, the underlying...
FDA-mandated CAR-T monitoring period could be halved, say researchers
In patients with diffuse large B-cell non-Hodgkin lymphoma (DLBCL), the two hallmark post-chimeric antigen receptor (CAR)-T therapy toxicities are extremely rare after two weeks, supporting a shorter, more flexible toxicity monitoring period, according to a study...
Ancestral CRISPR-Cas13 Ribonucleases Discovered: Implications for Genome Editing
In a pioneering study published in *Science*, a team of researchers led by Peter H. Yoon and Jennifer A. Doudna from the University of California, Berkeley, has made a remarkable discovery in the realm of CRISPR technology. The team has identified an ancestral clade...
KBI Biopharma Expands Manufacturing Contract with Global Pharmaceutical Company
KBI Biopharma Inc., a JSR Life Sciences company and global cGMP contract development and manufacturing organization (CDMO), has extended and expanded its manufacturing contract with a leading global pharmaceutical company. Originally initiated in 2020, the renewed...
Related Services

Plasmids GMP Services
Multiple scales & grade of solutions of various kind of plasmids suitable for multiple treatments in a fast and cost effective way.
READ MORE

AAV GMP Services
Ranging from small-scale AAV production, to large-scale AAV cGMP manufacturing for animal studies.
READ MORE

Technology Platforms
PackGene’s proprietary π-Alpha™ 293 AAV High-yield Platform increases AAV production by 3 to 8 times that of traditional platforms.
READ MORE

