A team of researchers has used lipid nanoparticles loaded with mRNA—the starring technology in some COVID-19 vaccines—to noninvasively and selectively trigger cell death in living mice’s blood stem cells. And in a second experiment, they used the nanoparticle system to remove a sickle cell–producing gene in human cells (Science 2023; DOI: 10.1126/science.ade6967).
Several therapies that involve modifying blood cell precursors in the bone marrow called hematopoietic stem cells (HSCs) already exist. But they involve extracting the HSCs, manipulating them in a sterile environment, and returning them to the body—a cumbersome and expensive process. And often, before receiving the modified HSCs, people must go through a regimen of toxic chemotherapy to kill off the ones already in the bone marrow.
The new advance involves a single injection and uses antibodies to target the payload to its intended destination. And it makes its cellular or genetic modifications directly in the body without the need for cell extraction.
The researchers developed two payloads: one that edited a mutation for sickle cell disease, and another that selectively killed HSCs, which would eliminate the need for chemotherapy before HSC transplantion. If the therapies can be successfully adapted to people, this approach “will actually make gene therapy affordable, not only to our patients but also to our health care system,” says Hamideh Parhiz, a biotechnologist at the University of Pennsylvania, who co-led the research.
The researchers designed the lipid nanoparticles to target HSCs using an antibody that binds to the protein CD117, which is commonly found on these cells’ surface. After confirming that the nanoparticles were breaking through into about half of blood cells, they loaded the antibody-coated nanoparticles with an mRNA encoding a protein that induces cell death. Although the nanoparticles killed HSCs, the researchers discovered some off-target effects, so they added tiny bits of noncoding RNA that kept the protein from killing other cells. “That’s when we got success,” Parhiz says.
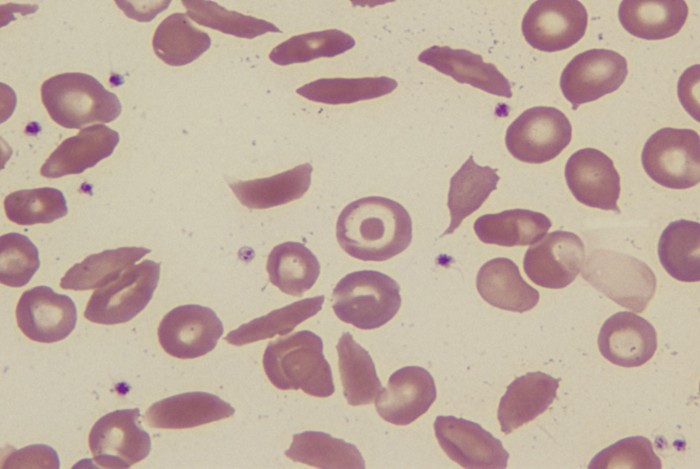
In another experiment, the researchers stuffed their nanoparticles with an mRNA sequence that produces a gene editor when it enters the cell. The editor targets a mutation in hemoglobin causing sickle cell disease.
The researchers tested the gene-editing nanoparticles on cells grown from samples taken from people with the disease. Reversing the mutation resulted in more than 95% of blood cells taking on a typical round shape rather than the sickle-like appearance characteristic of the disease.
Parhiz and her colleagues are working on fine-tuning the approach and testing it further in animals to get a better understanding of how efficiently it edits intended genes and how well it targets HSCs.
The study is “an impressive advance,” says David R. Liu, a chemist and gene editing expert at the Broad Institute of MIT and Harvard. Though many steps remain before clinical testing, he says, the approach “could lay a foundation for the much broader availability of programmable therapeutic gene editing to treat a variety of genetic blood disorders, which would represent a major breakthrough for patients.”

Check out our mRNA service to expedite your vaccine research
PackGene is a CRO & CDMO technology company that specializes in packaging recombinant adeno-associated virus (rAAV) vectors. Since its establishment in 2014, PackGene has been a leader in the AAV vector CRO service field, providing tens of thousands of custom batches of AAV samples to customers in over 20 countries. PackGene offers a one-stop CMC solution for the early development, pre-clinical development, clinical trials, and drug approval of rAAV vector drugs for cell and gene therapy (CGT) companies that is fast, cost-effective, high-quality, and scalable. Additionally, the company provides compliant services for the GMP-scale production of AAVs and plasmids for pharmaceutical companies, utilizing five technology platforms, including the π-Alpha™ 293 cell AAV high-yield platform and the π-Omega™ plasmid high-yield platform. PackGene’s mission is to make gene therapy affordable and accelerate the launch of innovative gene drugs. The company aims to simplify the challenging aspects of gene therapy development and industrialization processes and provide stable, efficient, and economical rAAV Fast Services to accelerate gene and cell therapy development efforts from discovery phase to commercialization.
Related News
Exploring Tau Protein’s Role in Glaucoma: New Insights and Therapeutic Potential
Glaucoma, a chronic neurodegenerative disorder, leads to irreversible vision loss by damaging retinal ganglion cells (RGCs) and the optic nerve, often associated with increased intraocular pressure (IOP). Despite the benefits of IOP-lowering treatments, the underlying...
FDA-mandated CAR-T monitoring period could be halved, say researchers
In patients with diffuse large B-cell non-Hodgkin lymphoma (DLBCL), the two hallmark post-chimeric antigen receptor (CAR)-T therapy toxicities are extremely rare after two weeks, supporting a shorter, more flexible toxicity monitoring period, according to a study...
Ancestral CRISPR-Cas13 Ribonucleases Discovered: Implications for Genome Editing
In a pioneering study published in *Science*, a team of researchers led by Peter H. Yoon and Jennifer A. Doudna from the University of California, Berkeley, has made a remarkable discovery in the realm of CRISPR technology. The team has identified an ancestral clade...
KBI Biopharma Expands Manufacturing Contract with Global Pharmaceutical Company
KBI Biopharma Inc., a JSR Life Sciences company and global cGMP contract development and manufacturing organization (CDMO), has extended and expanded its manufacturing contract with a leading global pharmaceutical company. Originally initiated in 2020, the renewed...
Related Services

Plasmids GMP Services
Multiple scales & grade of solutions of various kind of plasmids suitable for multiple treatments in a fast and cost effective way.
READ MORE

AAV GMP Services
Ranging from small-scale AAV production, to large-scale AAV cGMP manufacturing for animal studies.
READ MORE

Technology Platforms
PackGene’s proprietary π-Alpha™ 293 AAV High-yield Platform increases AAV production by 3 to 8 times that of traditional platforms.
READ MORE

