Here are two things that are true.
The world needs more effective flu vaccines. And pharmaceutical companies that learned of the vaccine-making power of the messenger RNA platform during the Covid-19 pandemic need new markets for their technology.
With the heydays of Covid vaccine sales in the rearview mirror, the flu vaccine market, with its antiquated production process that mainly relies on growing viruses in hen’s eggs, seems an obvious candidate for a shake-up.
The manufacturers have noticed, with Pfizer, Moderna, GSK, and Sanofi all working on mRNA flu shots. GSK and Sanofi are already major producers of flu vaccines and may be looking to solidify their positions; Pfizer and Moderna are new to flu, but further along in the mRNA work.
Given the widespread acceptance that the current flu vaccines could use improvement, are mRNA shots the answer? As the scientific world waits for published data on which to formulate conclusions, STAT spoke to influenza and vaccine experts who see some significant benefits of applying mRNA technology to the production of flu vaccine — but also some serious challenges mRNA manufacturers will face breaking into this already crowded market.
Let’s explore the arguments for and against mRNA flu shots.
Advantage: Flu shots might not miss the mark as often
The major challenge with flu vaccines is that to make them involves predicting the future.
Flu viruses change constantly. As a result, experts meet twice a year at the World Health Organization to pore over global data, looking at which strains of the viruses — H1N1, H3N2, and two types of influenza B — seem to be picking up speed and which appear to be losing ground. In late February they choose the four they think will be the biggest threat in the following Northern Hemisphere season; in late September, they do the same for the next Southern Hemisphere winter. (This number will soon shrink to three; the WHO has urged manufacturers to remove the influenza B/Yamagata component from flu vaccines because those viruses no longer seem to be circulating.)
The choices — educated guesses, really — must be made far in advance because it takes manufacturers nearly half a year to produce the vaccine, bottle and label it, and distribute doses to doctors offices, workplace clinics, and pharmacies. Some years the selected strains protect well against what is making the rounds. Other years, they don’t.
Vaccine production using the mRNA platform is quicker; for one thing, it doesn’t involve growing viruses. So vaccine strains could be selected closer to the actual season, potentially reducing the likelihood of mismatches. We saw this advantage in action when the Food and Drug Administration selected the Covid strain targeted in this fall’s shot. It was chosen in mid-June; the vaccine rollout began in mid-September.
Flu vaccine might take longer to make, because the vaccine targets multiple viruses. Still, Raffael Nachbagauer, head of Moderna’s flu vaccine program, said he wouldn’t rule out the possibility that flu vaccine could be made on that timeline. An alternative might be a rolling strain selection process. “Maybe you don’t need to decide on all three strains in June, for example. Maybe you feel quite certain in April about the B strain, because the B strain hasn’t been changing as much,” he said.
Choosing vaccine strains closer to flu season would be a huge advantage, said Florian Krammer, an influenza virologist at the Icahn School of Medicine at Mount Sinai. “If you can pick your strains after you know what’s mainly circulating in the Southern Hemisphere [winter], I think you’d have a much bigger chance to have a matched vaccine.”
Scott Hensley, a professor of microbiology at the University of Pennsylvania’s Perelman School of Medicine, said the mRNA platform might help improve the flu shot in another way. In the current egg-based manufacturing process, which most producers use, viruses can acquire deleterious mutations as they are grown in eggs. The process, known as egg adaptation, can effectively knock the vaccines off target. They train our immune systems to protect against viruses that may have important differences from the ones making people sick, creating a mismatch. This wouldn’t be an issue for mRNA flu vaccines, which do not contain killed viruses.
Advantage: A foundation for quick vaccines in the next flu pandemic
The biggest advantage of producing mRNA flu shots doesn’t relate to seasonal vaccine supplies, however. Where this technology would really shine would be in the event of a flu pandemic.
When a flu pandemic starts, vaccine manufacturing operations pivot to making vaccines to target the new influenza strain. But the current global output isn’t adequate to make the doses we’d need in a flu pandemic.
“Our capacity for responding to a [flu] pandemic using egg-based or even cell-based vaccines is … about a billion doses a year,” said Barney Graham, an immunologist and virologist who teaches at Morehouse University School of Medicine, and one of the key figures involved in the design of Covid-19 vaccines. “That’s not enough for 7 billion people on Earth. So mRNA could help solve that supply problem.”
Even with the production capacity that exists, trying to grow a new virus in eggs can be a slower-than-optimal process, as was seen in the 2009 H1N1 pandemic. By the time enough doses were available to vaccinate in the United States, the big wave of infections here was well past its peak. Most of the world made it through the H1N1 pandemic without the help of vaccines.
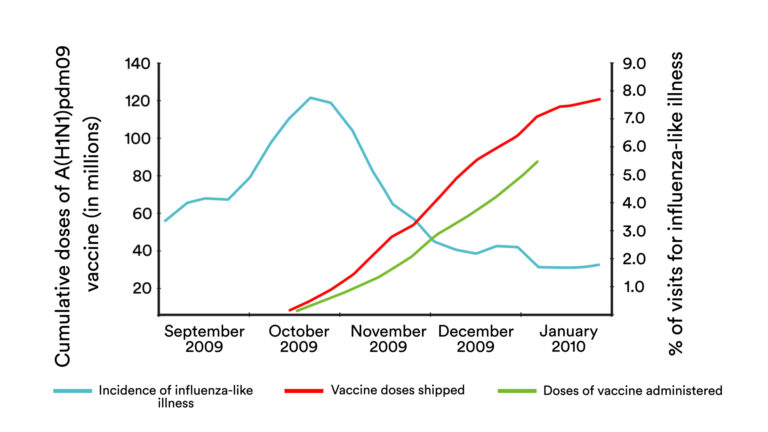
The 2009 pandemic was mild by historic standards. But in the event of a more severe pandemic, every day that could be shaved off the vaccine production timeline would save lives.
To take maximum advantage of that speed, however, we would have to have already licensed seasonal mRNA flu shots. That’s because of a quirk of flu vaccine regulation that is used to manage the need to constantly update the strains that flu shots target. The FDA and other major regulatory agencies do not require manufacturers to conduct large vaccine efficacy trials every time a component of the flu vaccine is updated; they do not consider the updated vaccine a new product.
The “strain change” rule applies in a pandemic as well, meaning that once the vaccine is made, it doesn’t have to undergo the large-scale efficacy trials that Covid shots did in 2020. Small trials would be needed to show that the vaccine was safe and to determine the effective dose, but after that, the vaccine could be deployed. The wait for mRNA flu vaccines in a pandemic could be months shorter than what we experienced in 2020.
But pandemics are rare and unpredictable events. The last four occurred in 1918, 1957, 1968, and 2009. And while government pandemic planners count on seasonal flu production capacity to respond to pandemics, manufacturers rely on seasonal flu vaccine sales to stay in the market.
“Manufacturing advantages aren’t necessarily market advantages most years,” noted Kathleen Neuzil, director of the University of Maryland School of Medicine’s Center for Vaccine Development and Global Health.
Challenge: The mRNA baggage
Some people continue to get Covid shots whenever they are recommended by health authorities. But public acceptance of Covid boosters has declined with each successive campaign. Only 16% of adults — and 32% of adults aged 65 and older — have received this fall’s shot so far, according to the latest data from the Centers for Disease Control and Prevention. Flu shot uptake this year is about double that.
Covid fatigue is definitely playing a role. But some of the poor uptake is due to a stubborn distrust of the mRNA technology within a portion of the population.
“I think there are data to support the fact that there is a certain segment of the population that is extremely reluctant to get an mRNA vaccine,” said Michael Osterholm, director of the University of Minnesota’s Center for Infectious Disease Research and Policy, which earlier this year published a roadmap for development of better influenza vaccines.
Even the best vaccine is only effective if it’s accepted by the people you hope to protect, he said. “If you don’t get it, you’ve got a [vaccine] effectiveness of zero.”
Challenge: The side effect profile
When mRNA Covid vaccines became available in the winter and spring of 2021, people started swapping side effect stories. Some felt nothing, or reported at most a bit of a sore arm. Some spiked high fevers, experienced chills, aches, malaise — symptoms that approximate the experience of having the flu — for a day or two. Some get knocked flat every time they get an mRNA shot.
“I don’t react,” said Arnold Monto, an influenza epidemiologist at University of Michigan School of Public Health. “My daughter … knows she can’t be doing any normal activities for about 48 hours.”
Traditional flu shots are not associated with the side effects that mRNA shots have become known for.
That reputation for reactogenicity, as the the likelihood of unpleasant side effects is called, will probably turn some people off of the idea of mRNA flu shots, said Ben Cowling, chair of epidemiology at Hong Kong University’s School of Public Health. “With the reactogenicity profile, with the likely cost, I’m not sure what the uptake would be.”
Challenge: Cost
Traditional flu shots are cheap, especially compared to some of the new entrants in the overall vaccines market. Take GSK’s and Pfizer’s new RSV vaccines for seniors. They cost $280 and $295, respectively, if paid for by the private sector.
Makers of mRNA vaccines won’t be able to charge those kinds of premiums for flu shots. “We have many options,” said Neuzil. “So any new influenza vaccine has to have a competitive advantage.”
Regular flu shots cost between about $15 and $30, depending on whether the purchaser is the federal government or a private insurance plan. A few vaccines — two made by Sanofi, one by Seqirus — are sold at premium prices, around $75 a dose, because they have been shown to generate more protection in older adults. That enhanced protection earned them what’s known as a preferential recommendation from the Advisory Committee on Immunization Practices, an expert panel that helps the CDC form its vaccine guidance. If you’re 65 or older, you’ll get one of those vaccines.
Experts watching the work on mRNA flu shots think it’s unlikely Pfizer and Moderna are looking to sell cheap mRNA flu shots. These are the companies, after all, that effectively quadrupled the prices of their Covid shots this fall when the vaccines were sold on the open market for the first time. Moderna now charges about $82 when it sells to government programs and $128 to the private sector; Pfizer’s prices have gone to $85 and $115 respectively, according to the CDC.
But unless mRNA flu shots can earn a preferential recommendation from ACIP, it’s hard to see how companies could charge Covid-shot type prices — none of the flu vaccine manufacturers currently earns those — and still snag a share of an already crowded flu shot market. “Clearly if they are going to be coming with a vaccine at four times the cost, they’re going to have to at least show twice the benefit,” Monto said.
To score a preferential recommendation, mRNA flu shot makers will have to prove that their vaccines are more effective than regular flu shots, and at least as effective as the enhanced shots now recommended for seniors.
In September, Moderna reported to shareholders that its mRNA flu shot induced higher levels of antibodies than Sanofi’s high-dose vaccine, Fluzone. Nachbagauer said Moderna hopes to have its mRNA flu vaccine licensed using that immunogenicity data, and would follow up with an effectiveness trial post-licensure.
Pfizer is in the late stages of a Phase 3 trial and expects to have topline results soon. But it is testing its mRNA flu vaccine against a regular flu shot, not an enhanced vaccine.
Influenza experts are keen to see published data from these trials.
“There’s press releases from Moderna. There’s no data from Pfizer that’s publicly available. So we can only speculate. But it doesn’t look like what you get with the mRNA vaccines … is much better than what you get with regular seasonal flu vaccines,” said Krammer.
Graham believes there could be additional benefits from the mRNA vaccines, because they draw a part of the immune system not activated by existing flu shots, CD8+ T cells, into the fight. That could help people who become infected to recover faster, in the process cutting back on the amount of flu spreading from person to person. “Shorter transmission, shorter viral loads for a shorter period of time. So it might have an effect on transmission. But I think that would have to be determined,” Graham said.
Challenge: The market is already crowded
The United States has what’s known as a universal recommendation for flu vaccination; the CDC says that everyone over the age of six months should get a flu shot every year.
Though only about half of the country follows that advice, the recommendation has created a large market for flu vaccine. In recent years the combined U.S. purchases from both government and private buyers has been in the range of 170 million to 175 million doses, supplied by GSK, Sanofi, Seqirus, and AstraZeneca. (The latter sells only the needleless vaccine FluMist, which is puffed into a nostril.)
Finding a niche in a crowded market isn’t going to be easy, said Norman Baylor, a former director of FDA’s office of vaccines who now runs Biologics Consulting. “I don’t see it at this time, unless they can demonstrate an advantage over what we already have,” he said.
Advantage? Challenge? The jury is still out on combo vaccines
Pfizer, Moderna, GSK, and Sanofi are all working on mRNA flu vaccines. While the latter two companies appear to be focused strictly on standalone flu shots, Pfizer and Moderna are both exploring the possibility of combining flu and Covid shots, and flu and RSV shots; Moderna is even working on an experimental vaccine that would protect against flu, Covid, and RSV.
If those combination vaccines work, that could provide an entree to the market and premium prices. But a critical question hangs over these projects: Do RSV and Covid vaccines need to be given with the same frequency as flu shots? From a marketing point of view, the combination shots really only work if people need to be vaccinated against the viruses they target every year.
“For influenza and Covid, the question to me that hasn’t been completely settled is, is Covid a seasonal virus that matches influenza?” Neuzil said. “I think we all hope it’s going to get there … [but] I’m not sure it’s entirely settled into that seasonal pattern yet.”
And she noted that the rapidly waning immunity from Covid shots has led health authorities to allow older adults and people who are immunocompromised to get two Covid shots a year for the past two years. The ACIP has already said immunocompromised people can get a second Covid shot this winter and will study if people over 65 should too. If this twice-a-year Covid shot pattern for high-risk people holds, a combination shot could only be given in the autumn; a solo Covid booster would need to be given some months later.
There are questions too about how often people will need to be revaccinated against RSV. Both GSK and Pfizer, which have brought RSV vaccines for older adults to market, have indicated their studies show that the protection lasts at least 18 months (Pfizer) and 24 months (GSK). (Their studies are ongoing; protection may last longer still.) If a vaccine isn’t needed annually, it wouldn’t make a good candidate for inclusion in an annual shot.
“That’s premature,” Cowling said of the idea of combining RSV vaccine with flu shots.
Challenge: The unfulfilled promise
All involved in influenza research would love to see flu vaccines that would generate longer-lasting and broader protection, so that flu viruses wouldn’t be able to so easily evade the immunity vaccines induce. Or vaccines like one Hensley is working on, aimed at giving people some protection against 20 subtypes of flu that don’t currently circulate in people but may pose a pandemic threat in the future.
The mRNA platform could be the right one for designing such vaccines, some experts believe. But that would involve more research and the creation of new vaccines that would need to prove to regulators that they can actually achieve things the existing products do not. That would be a longer path to the market than the approach manufacturers are currently taking, which is to produce a product similar to existing shots, just made in a different manner.
Moderna is testing experimental flu vaccines that include another part of flu viruses, theorizing that inclusion of a protein called the neuraminidase could broaden protection; Krammer thinks this approach could help lessen the impact of vaccine mismatches. Nachbagauer said Moderna is mulling over what evidence might persuade regulatory agencies to license such a product.
The company is also exploring adding an additional H3N2 virus to a seasonal vaccine, using the space that will be vacated by the removal of the B/Yamagata component. H3 viruses change more rapidly than the others and are more likely to be mismatched in the vaccine. The work, at this point, is to establish proof of concept, Nachbagauer said. “It’s essentially a conversation starter on ‘Is this possible?’ and then ‘How do you go from there?’”
Krammer, for one, thinks using the mRNA platform to produce only status-quo flu vaccines would be almost tragic. “I think the mRNA platform has so much flexibility and it would be really shortsighted to just push for something that’s equivalent to what we have right now, instead of pushing for something that’s better.”

Check out our mRNA service to expedite your vaccine research
PackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span from vector design and construction to AAV, lentivirus, and mRNA services. With a sharp focus on early-stage drug discovery, preclinical development, and cell and gene therapy trials, we deliver cost-effective, dependable, and scalable production solutions. Leveraging our groundbreaking π-alpha 293 AAV high-yield platform, we amplify AAV production by up to 10-fold, yielding up to 1e+17vg per batch to meet diverse commercial and clinical project needs. Moreover, our tailored mRNA and LNP products and services cater to every stage of drug and vaccine development, from research to GMP production, providing a seamless, end-to-end solution.
Related News
Exploring Tau Protein’s Role in Glaucoma: New Insights and Therapeutic Potential
Glaucoma, a chronic neurodegenerative disorder, leads to irreversible vision loss by damaging retinal ganglion cells (RGCs) and the optic nerve, often associated with increased intraocular pressure (IOP). Despite the benefits of IOP-lowering treatments, the underlying...
FDA-mandated CAR-T monitoring period could be halved, say researchers
In patients with diffuse large B-cell non-Hodgkin lymphoma (DLBCL), the two hallmark post-chimeric antigen receptor (CAR)-T therapy toxicities are extremely rare after two weeks, supporting a shorter, more flexible toxicity monitoring period, according to a study...
Ancestral CRISPR-Cas13 Ribonucleases Discovered: Implications for Genome Editing
In a pioneering study published in *Science*, a team of researchers led by Peter H. Yoon and Jennifer A. Doudna from the University of California, Berkeley, has made a remarkable discovery in the realm of CRISPR technology. The team has identified an ancestral clade...
KBI Biopharma Expands Manufacturing Contract with Global Pharmaceutical Company
KBI Biopharma Inc., a JSR Life Sciences company and global cGMP contract development and manufacturing organization (CDMO), has extended and expanded its manufacturing contract with a leading global pharmaceutical company. Originally initiated in 2020, the renewed...
Related Services

