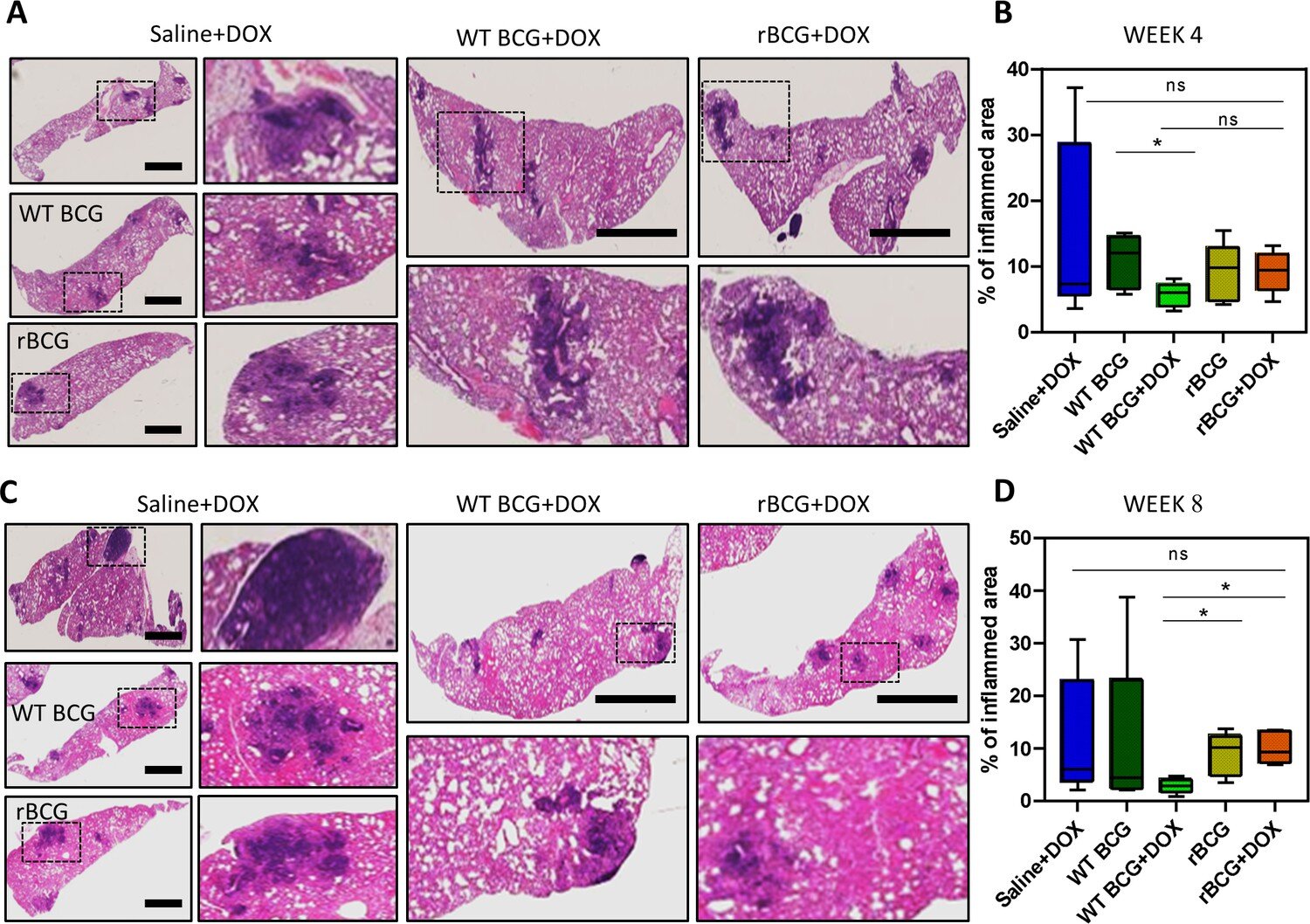
Histopathological analysis of lung samples. (A) Histological haematoxylin and eosin (H&E) staining of lung samples at week 4 post Mtb challenge. (B) Analysis of percentage of inflamed area (indicated with black boxes) from each mouse lung per immunized group. (C) H&E staining of lung samples at week 8 post Mtb H37Rv infection. (D) Analysis of percentage of inflamed area from each mouse lung. Credit: eLife (2024). DOI: 10.7554/eLife.89157
In a critical global public health development, a candidate vaccine for tuberculosis (TB) has been created using a gene-editing approach.
TB remains the leading cause of death by infectious disease globally, with South Africa having one of the highest incidence rates in the world.
While the BCG vaccine used to prevent TB is widely available for infants, no vaccine has shown lasting protection. The BCG is also the only existing effective vaccine.
“South Africa committed to the Sustainable Development Goal of ending the TB epidemic by 2030. While we are doing relatively well as a country—TB deaths have come down since 2015—we need to do a lot better to reach the milestones,” says Professor Bavesh Kana.
Kana, the Head of the School of Pathology and former director of the Center of Excellence for Biomedical TB Research at Wits University, contributed to the new study published in eLife.
The researchers modified the BCG vaccine to make it more effective at controlling the growth of M. tuberculosis. Mice injected with the edited BCG vaccine had less M. tuberculosis growth in their lungs than mice that received the original vaccine.
“We can now offer a new candidate vaccine in the fight against this deadly disease,” says Kana. “The work also demonstrates that gene editing is a powerful way to develop vaccines. This is particularly important for researchers working on vaccine development.”
About the tuberculosis vaccine
The BCG vaccine is given to children around the time of birth and is effective at preventing TB disease. However, BCG does not protect teenagers and adults and has not been effective at eradicating TB.
This has spurred the need to develop novel TB vaccine candidates to replace or boost BCG.
“We also see that the BCG can evade the immune system and that this reduces its efficacy as a vaccine,” says Kana. He noted that the importance of vaccines cannot be overstated.
When humans get sick, the body’s defense system spots particular signs, called PAMPs (pathogen-associated molecular patterns), on the outside of bacteria, viruses, or other harmful germs.
This helps the body tell the difference between invaders and its own cells and then starts fighting the infection.
Vaccines work by looking like germs, so that they can start the first defense without making a person sick.
Kana has lamented the funding gap in developing tools to eliminate TB—a disease which dates back over 9,000 years. “Until recently, our diagnostic approaches were a century old. With some novel vaccine candidates in the pipeline, we can finally begin to adequately address this devastating illness.”
https://medicalxpress.com/news/2024-06-potential-vaccine-tuberculosis-people-ages.html

Check out our AAV CDMO service to expedite your gene therapy research
PackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span from vector design and construction to AAV, lentivirus, and mRNA services. With a sharp focus on early-stage drug discovery, preclinical development, and cell and gene therapy trials, we deliver cost-effective, dependable, and scalable production solutions. Leveraging our groundbreaking π-alpha 293 AAV high-yield platform, we amplify AAV production by up to 10-fold, yielding up to 1e+17vg per batch to meet diverse commercial and clinical project needs. Moreover, our tailored mRNA and LNP products and services cater to every stage of drug and vaccine development, from research to GMP production, providing a seamless, end-to-end solution.
Related News
Sangamo Therapeutics Secures Accelerated Approval Pathway for Gene Therapy in Fabry Disease
Sangamo Therapeutics has announced a major advancement in its gene therapy program for Fabry disease, as the U.S. FDA has provided a clear pathway for Accelerated Approval. This decision could potentially speed up approval timelines by three years, with a Biologics...
[2024/10/18] Gene and Cell Therapy- weekly digest from PackGene
FeaturedNewsArticlesPackGene's NewsletterReceive the latest news and insights to your inbox.About PackGenePackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span...
GSK sues Moderna over mRNA vaccine patents, seeks ‘reasonable royalty’
GSK on Tuesday unveiled a lawsuit filed against Moderna in Delaware federal court, alleging that its patented inventions provide the “foundation” for Moderna’s mRNA vaccine portfolio. GSK said it’s looking to recover “a reasonable royalty” for Moderna’s tens of...
Gene Therapy Automatically Converts Omega-6 to Omega-3 Fatty Acids in the Body
Shriners Children's Develops New Technology to Prevent Childhood Obesity ST. LOUIS, Oct. 16, 2024 /PRNewswire/ -- According to the Centers for Disease Control, nearly 20% of children and teens are considered obese. Research shows it can have a dramatic impact on a...
Related Services

Plasmids GMP Services

AAV GMP Services


