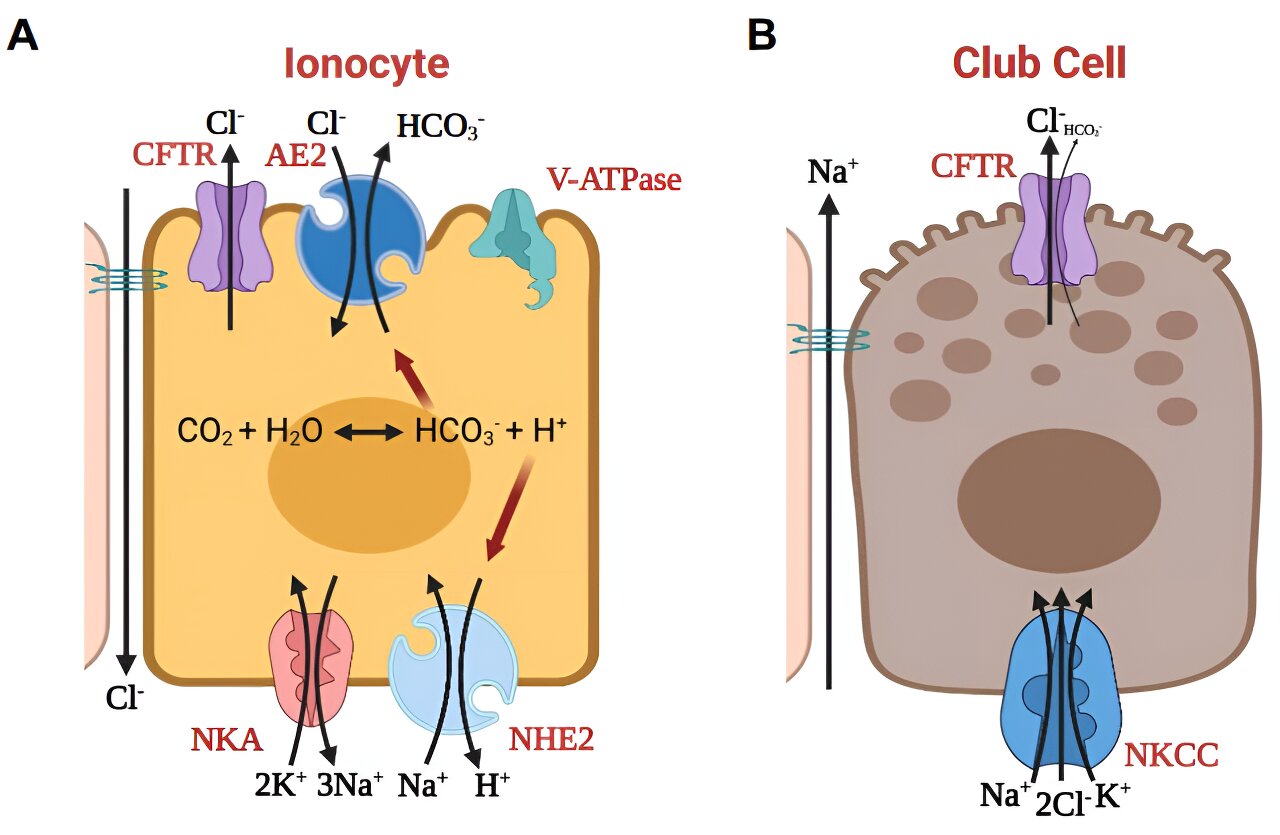
Schematic model of working hypothesis of how (A) pulmonary ionocytes and (B) club cells function in pHBE cell culture stimulated with forskolin plus IBMX. Credit: American Journal of Respiratory and Critical Care Medicine (2024). DOI: 10.1164/rccm.202309-1565OC
A team of University of Saskatchewan (USask) researchers are exploring the role of a newly identified cell type in cystic fibrosis (CF), which could lead to effective new types of treatment.
Researchers led by the College of Medicine’s Dr. Juan Ianowski (Ph.D.) in the Department of Anatomy, Physiology and Pharmacology, and Dr. Julian Tam (MD) in the Division of Respirology, Critical Care and Sleep Medicine, recently published a paper in the American Journal of Respiratory and Critical Care Medicine highlighting the function of pulmonary ionocyte cells in CF.
CF disease is a genetic condition caused by mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) protein. Because some potential treatments of CF involve targeting and modifying the CFTR function at the genetic level in sick airway cells, the specific role various types of cells play in CF disease—like the pulmonary ionocyte—is critical for creating targeted treatments. This newly identified cell type is now being explored by Ianowski and Tam’s team of researchers.
“We do have a lot to offer when it comes to testing and experimental models,” Ianowski said. “Gene therapy treatments, if they work and we have a systematic way of developing them, it’s a game changer for this and other diseases as well.”
Pulmonary ionocytes, a recently discovered and rare lung cell type, were identified as existing in small numbers—fewer than one percent of the airway surface cells in the lungs. Ianowski and Tam’s research suggests that these cells play a crucial role in the pH levels of airway surface liquid (ASL).
“It’s been shown that abnormal airway acidity is an important component of CF lung disease pathobiology,” Tam said. “What we found is that pulmonary ionocytes regulate the acidity of CF airways.”
CF disease affects cells throughout the body and the glands that produce sweat and mucous. Some of the symptoms of CF disease include thickened mucous, difficulty clearing the airways of mucous, and susceptibility to lung infections and other similar issues.
ASL with high acidity has been shown to increase the difficulty of mucociliary clearance and reduce resistance to infection—which means pulmonary ionocytes could be a key factor in how CF disease manifests.
“This minute population of cells express most of the RNA for CFTR,” Ianowski said. “So now there is a paradigm shift for the field … ‘Is it possible to tag those cells? How easy is it going to be to fix those cells, and also, with what?’ It’s a very difficult thing to study because how do you study 0.5% of the entire epithelia?”
Research into this new cell is still ongoing. Ianowski noted that the role and distribution of pulmonary ionocytes do not appear to be random, but the reason and function behind their distribution are not completely understood yet.
This new type of cell has been a focus of CF research around the world since its discovery. While some research has suggested that pulmonary ionocytes play a role in the creation of ASL, Tam and Ianowski’s research suggests it affects the acidity of the liquid only.
Better comprehension of the function of the cell—and how that function relates to symptoms of CF—will help support future treatments.
“One of our lab’s tools is a specialized technique that allows us to study cells at a single-cell level,” Tam said. “We think it’s important to correct the appropriate cells for gene therapies, so having an understanding of which cells have which function would allow researchers to do this.”
Ianowski said he’s excited for new opportunities to collaborate with other researchers in the development of gene therapies to combat CF. While this research team might not be the ones to create those treatments, Ianowski said their unique lab at USask is well-suited to provide the underlying expertise—like their work analyzing the function of pulmonary ionocytes—to help inform the creation of advanced CF therapies in the future.
https://medicalxpress.com/news/2024-04-explore-cell-cystic-fibrosis-treatment.html
PackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span from vector design and construction to AAV, lentivirus, and mRNA services. With a sharp focus on early-stage drug discovery, preclinical development, and cell and gene therapy trials, we deliver cost-effective, dependable, and scalable production solutions. Leveraging our groundbreaking π-alpha 293 AAV high-yield platform, we amplify AAV production by up to 10-fold, yielding up to 1e+17vg per batch to meet diverse commercial and clinical project needs. Moreover, our tailored mRNA and LNP products and services cater to every stage of drug and vaccine development, from research to GMP production, providing a seamless, end-to-end solution.
Related News
Sangamo Therapeutics Secures Accelerated Approval Pathway for Gene Therapy in Fabry Disease
Sangamo Therapeutics has announced a major advancement in its gene therapy program for Fabry disease, as the U.S. FDA has provided a clear pathway for Accelerated Approval. This decision could potentially speed up approval timelines by three years, with a Biologics...
[2024/10/18] Gene and Cell Therapy- weekly digest from PackGene
FeaturedNewsArticlesPackGene's NewsletterReceive the latest news and insights to your inbox.About PackGenePackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span...
GSK sues Moderna over mRNA vaccine patents, seeks ‘reasonable royalty’
GSK on Tuesday unveiled a lawsuit filed against Moderna in Delaware federal court, alleging that its patented inventions provide the “foundation” for Moderna’s mRNA vaccine portfolio. GSK said it’s looking to recover “a reasonable royalty” for Moderna’s tens of...
Gene Therapy Automatically Converts Omega-6 to Omega-3 Fatty Acids in the Body
Shriners Children's Develops New Technology to Prevent Childhood Obesity ST. LOUIS, Oct. 16, 2024 /PRNewswire/ -- According to the Centers for Disease Control, nearly 20% of children and teens are considered obese. Research shows it can have a dramatic impact on a...
Related Services

Plasmids GMP Services

AAV GMP Services


