Society learned about the value of mRNA during the COVID-19 pandemic when we saw scientists and medical professionals harness its power to deliver a vaccine for the virus within a year.
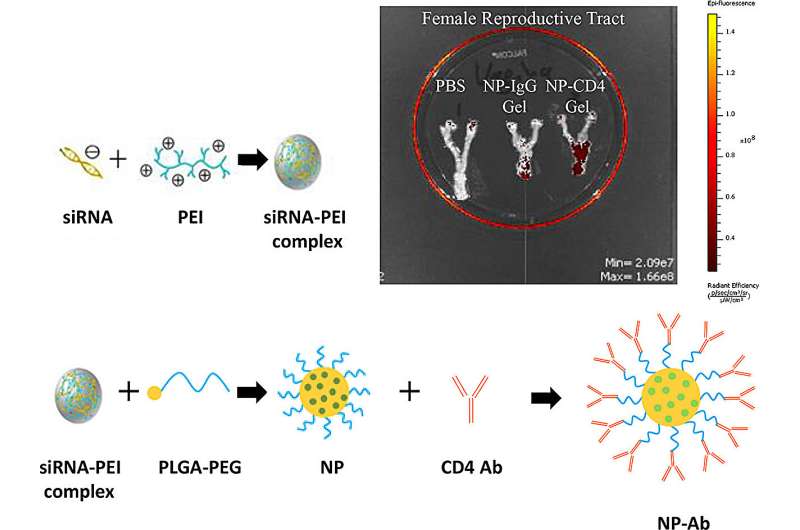
Now, University of Waterloo pharmacy associate professor Emmanuel Ho has developed a novel nanomedicine loaded with genetic material called small interfering RNAs (siRNA) to fight human immunodeficiency virus (HIV) using gene therapy. These siRNAs regulate which genes or proteins are turned on or off in our cells and showed a 73% reduction in HIV replication.
The study, “pH-sensitive dual-preventive siRNA-based nanomicrobicide reactivates autophagy and inhibits HIV infection in vaginal CD4+ cells,” was published in the Journal of Controlled Release.
“This opens the door for new therapeutics in the fight against HIV,” said Dr. Ho, who is among Waterloo’s researchers and entrepreneurs leading health innovation in Canada.
Autophagy, also known as the body’s recycling process, plays an important role in our body to eliminate microbes such as viruses and bacteria inside cells. HIV is quite smart and produces a protein, Nef, that prevents cells from activating autophagy.
This is the first research to develop a combination nanomedicine that can reactivate autophagy and prevent HIV entry into cells, allowing our body to re-initiate its defense system.
Additionally, HIV has a gene, CCR5, that allows the virus to enter a cell. The siRNAs target both Nef and CCR5 to reduce HIV infection.
This nanomedicine is intended to be applied vaginally to protect against sexual transmission of HIV. As a result, the nanomedicine is designed to be stable without leakage of siRNAs in the acidic vaginal environment but release the siRNA once inside cells.
“Viruses are smart. They produce Nef proteins to prevent autophagy from occurring,” Ho said. “Our process allows our body to fight the viral infection without needing additional drugs,”
Ho confirms that the next steps include further optimizing the process and improving our understanding of how autophagy plays a role in how our cells protect us from viruses.
“We also hope this will shed some light to develop more alternative approaches to effectively reduce antimicrobial resistance,” Ho said.

Check out our mRNA service to expedite your vaccine research
PackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span from vector design and construction to AAV, lentivirus, and mRNA services. With a sharp focus on early-stage drug discovery, preclinical development, and cell and gene therapy trials, we deliver cost-effective, dependable, and scalable production solutions. Leveraging our groundbreaking π-alpha 293 AAV high-yield platform, we amplify AAV production by up to 10-fold, yielding up to 1e+17vg per batch to meet diverse commercial and clinical project needs. Moreover, our tailored mRNA and LNP products and services cater to every stage of drug and vaccine development, from research to GMP production, providing a seamless, end-to-end solution.
Related News
Sangamo Therapeutics Secures Accelerated Approval Pathway for Gene Therapy in Fabry Disease
Sangamo Therapeutics has announced a major advancement in its gene therapy program for Fabry disease, as the U.S. FDA has provided a clear pathway for Accelerated Approval. This decision could potentially speed up approval timelines by three years, with a Biologics...
[2024/10/18] Gene and Cell Therapy- weekly digest from PackGene
FeaturedNewsArticlesPackGene's NewsletterReceive the latest news and insights to your inbox.About PackGenePackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span...
GSK sues Moderna over mRNA vaccine patents, seeks ‘reasonable royalty’
GSK on Tuesday unveiled a lawsuit filed against Moderna in Delaware federal court, alleging that its patented inventions provide the “foundation” for Moderna’s mRNA vaccine portfolio. GSK said it’s looking to recover “a reasonable royalty” for Moderna’s tens of...
Gene Therapy Automatically Converts Omega-6 to Omega-3 Fatty Acids in the Body
Shriners Children's Develops New Technology to Prevent Childhood Obesity ST. LOUIS, Oct. 16, 2024 /PRNewswire/ -- According to the Centers for Disease Control, nearly 20% of children and teens are considered obese. Research shows it can have a dramatic impact on a...
Related Services

