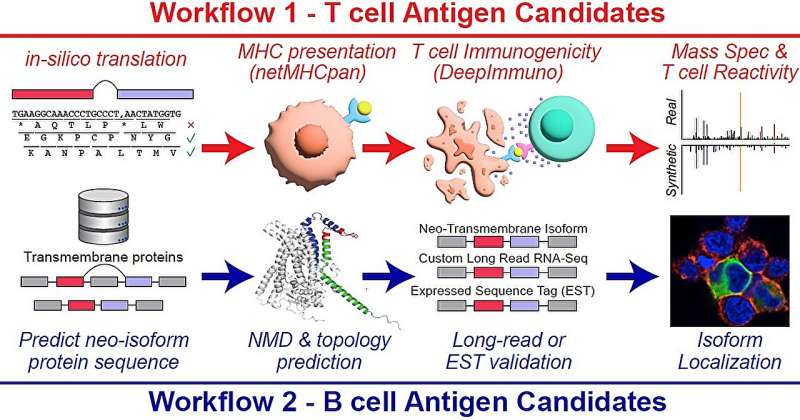
Researchers at Cincinnati Children’s outlined two workflows for detecting new targets for cancer immunotherapies, one based on T cell antigens, the other upon B cell antigens. Credit: Cincinnati Children’s
An innovative computational tool dubbed “SNAF” may help the research world bring the emerging promise of cancer immunotherapy to a wider range of patients, according to a study published in Science Translational Medicine.
The research tool, called the Splicing Neo Antigen Finder (SNAF), was developed by a multidisciplinary team of researchers from Cincinnati Children’s and the University of Virginia. The project was led by Guangyuan Li, Ph.D., and Nathan Salomonis, Ph.D., both with the Division of Biomedical Informatics at Cincinnati Children’s.
The co-authors say the tool has already helped uncover shared immunogenic targets across various cancers, which could pave the way for a new wave of highly focused cancer treatments.
“The implications of this discovery are significant,” says H. Leighton “Lee” Grimes, Ph.D., a co-author of the study and director of the Cancer Pathology Program at Cincinnati Children’s. “By identifying shared splicing neoantigens present in up to 90% of cancer patients, SNAF not only presents new targets for therapy but also challenges and expands our understanding of cancer biology.”
How the new tool finds new targets
Immunotherapy, which harnesses a patient’s immune system to fight cancer, often targets neoantigens produced from genetic mutations. However, this approach typically benefits only those with a high mutational burden. SNAF aims to expand the universe of immunotherapy by identifying neoantigens arising from post-transcriptional modifications, particularly splicing errors, which have been largely untapped until now.
Using new artificial intelligence approaches, SNAF predicts immunogenic peptides that T cells can recognize and novel proteins with altered extracellular components that B cells can target. This dual approach is vital in developing comprehensive immunotherapies that engage both arms of the adaptive immune system.
Promising target emerges for melanoma
While cataloging all possible neoantigens produced from alternative mRNA pathways, the team found that the amount of splicing neoantigens is correlated with patient survival and responses to immunotherapy in patients with melanoma.
One such prediction, SLC45A2, has emerged as a particularly promising target due to its high tumor specificity and immunogenicity.
In addition to T-cell neoantigens, the team has discovered a novel class of tumor-specific extracellular neo-epitopes, termed ExNeoEpitopes, through their B-cell focused pipeline, SNAF-B. These ExNeoEpitopes show great promise for the development of monoclonal antibodies and CAR-T cell therapies.
Next steps
“This is only the beginning,” says Tamara Tilburgs, Ph.D., co-corresponding author and a researcher in the Division of Immunobiology. “The SNAF workflow’s flexibility means it can be continuously adapted as we make further inroads into understanding and combating cancer.”
Salomonis and his team are now applying these tools in the most difficult-to-treat cancers to find the best targets for therapy development and understand their single-cell origins.
https://medicalxpress.com/news/2024-01-tool-cancer-immunotherapy.html
PackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span from vector design and construction to AAV, lentivirus, and mRNA services. With a sharp focus on early-stage drug discovery, preclinical development, and cell and gene therapy trials, we deliver cost-effective, dependable, and scalable production solutions. Leveraging our groundbreaking π-alpha 293 AAV high-yield platform, we amplify AAV production by up to 10-fold, yielding up to 1e+17vg per batch to meet diverse commercial and clinical project needs. Moreover, our tailored mRNA and LNP products and services cater to every stage of drug and vaccine development, from research to GMP production, providing a seamless, end-to-end solution.
Related News
Sangamo Therapeutics Secures Accelerated Approval Pathway for Gene Therapy in Fabry Disease
Sangamo Therapeutics has announced a major advancement in its gene therapy program for Fabry disease, as the U.S. FDA has provided a clear pathway for Accelerated Approval. This decision could potentially speed up approval timelines by three years, with a Biologics...
[2024/10/18] Gene and Cell Therapy- weekly digest from PackGene
FeaturedNewsArticlesPackGene's NewsletterReceive the latest news and insights to your inbox.About PackGenePackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span...
GSK sues Moderna over mRNA vaccine patents, seeks ‘reasonable royalty’
GSK on Tuesday unveiled a lawsuit filed against Moderna in Delaware federal court, alleging that its patented inventions provide the “foundation” for Moderna’s mRNA vaccine portfolio. GSK said it’s looking to recover “a reasonable royalty” for Moderna’s tens of...
Gene Therapy Automatically Converts Omega-6 to Omega-3 Fatty Acids in the Body
Shriners Children's Develops New Technology to Prevent Childhood Obesity ST. LOUIS, Oct. 16, 2024 /PRNewswire/ -- According to the Centers for Disease Control, nearly 20% of children and teens are considered obese. Research shows it can have a dramatic impact on a...
Related Services

Plasmids GMP Services

AAV GMP Services


