A Single, Easy Workflow: From Panel to NGS Report

1. Easy Ordering
Select your panel (Gen 1 vs. Gen 2) and choose your tissue add-ons. You can also add custom capsids.

2. Pooled Screening
Apply the single viral pool to your model (in vitro, organoid, or in vivo).

3. Harvest & Sequencing
Extract DNA and RNA and perform Next-Gen Sequencing (NGS). NGS reveals the distribution and functional expression of each capsid.

4. Bioinformatics
Receive a comprehensive report ranking the top performing capsids based on dual physical and functional transduction efficiency.
Key Benefits
-
High-Throughput & Unbiased
Screen over 99 novel and validated serotypes simultaneously, removing experimental variability by testing all variants in the same animal or dish. -
Dual DNA & RNA Readouts
Simultaneously quantify capsid entry via DNA and functional expression via RNA to prevent selecting capsids that enter the cell but fail to express. -
Versatile Application
Enjoy zero dropouts and an elite panel uniformity delivered in a ready-to-screen, pre-normalized pool.
Product Offerings & Workflow
We offer flexible screening options to suit your pipeline:
- Universal Panel (Generation 1): Includes ~70 serotypes (34 Novel CMRI, 12 PackGene, and 24 Published).
- Universal Panel (Generation 2): Includes ~99 serotypes (34 Novel CMRI, 12 PackGene, and 53 Published).
- Tissue-Specific Add-On Panels: Augment your panel with capsids pre-selected for specific target tissues.
- Custom Add-Ons: Add your own proprietary custom capsids to the panel.
Target Tissue Pre-Selected Serotypes CNS/Brain 21 serotypes Eye/Retina 9 serotypes Liver/Hepatocytes 5 serotypes Muscle & Heart 5 serotypes Kidney, Lung, T-Cells, and more Additional options available The entire single viral pool is applied to your model of choice, including in vitro cell cultures, organoids, and in vivo animal models. Next-generation sequencing (NGS) of the barcodes reveals the distribution and functional expression of each capsid.
Proven Performance Across In Vitro, Ex Vivo, and In Vivo Models
-
Our AAV Capsid Screening Kits consistently deliver accurate performance across various models, including in vitro (cell lines, primary cells, iPSCs), ex vivo (organoids), and in vivo (mouse, xenograft, NHP) systems. This is supported by rigorous quality control that ensures consistent and dependable data, enabling confident AAV selection for all downstream applications.
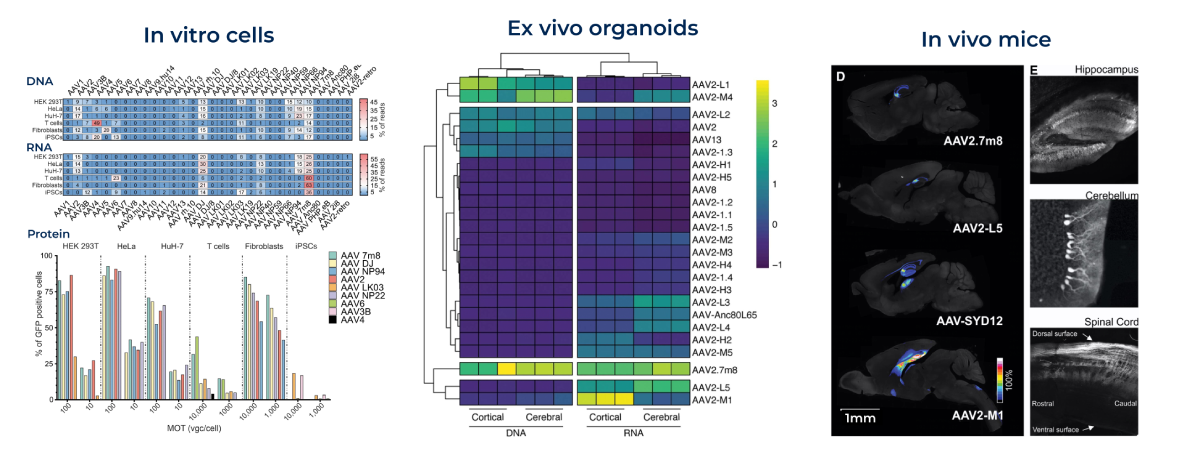
Westhaus et al. Human Gene Therapy. 2020. Drouyer et al. Molecular Therapy: Nucleic Acids. 2024.
Frequently Asked Questions
-
Q: What is the difference between the Generation 1 and Generation 2 Universal Panels?
A: The Universal Panel (Generation 1) includes approximately 70 pre-validated serotypes, featuring 34 novel CMRI capsids, 12 PackGene capsids, and 24 published capsids. The Generation 2 panel expands this panel to approximately 99 serotypes by including 53 published capsids alongside the novel options.
-
Q: How much do the screening kits cost?
-
Q: Can I add my own custom capsids to the screening pool?
-
Q: What tissue-specific panels are available?
-
Q: What experimental models are compatible with these kits?
-
Q: How does the barcoding system work for the dual readout?
-
Q: Is the kit compatible with single-cell RNA sequencing?
-
Q: How do you guarantee the uniformity and quality of the pooled library?
-
Q: Are the capsids in the pool perfectly equimolar, and do I need to normalize my data?
-
Q: What are the recommended storage conditions for the kits?
-
Q: Can I store the AAV Capsid Screening Kit at -80°C?
-
Q: Should I save a residual aliquot of the viral pool from my experiment?



