Key Advantages
-
Capacity
Our platform design results in higher library capacity -
Precision
Higher precision AAV-serotype screening -
Efficacy
High infectivity and tissue specificity can result in reduced dosage requirements that increase both safety and efficacy.
Intellectual Property
-
PackGene has three patents pending.
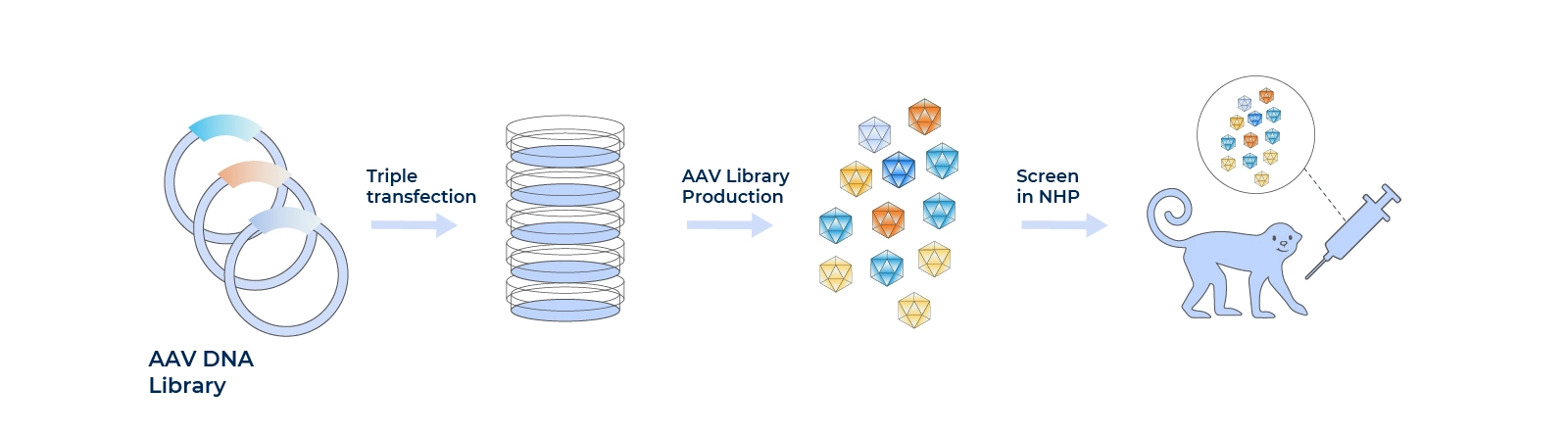
Screen the tissue-targeting AAVs to improve the efficiency
Library>1 billion mutants
Screens AAVs for tissue specificity.