
AAV Gene Therapy in Neurological Disorders From Neuroscience Research to Clinical Translation
Introduction
Neurological disorders affect billions worldwide and pose major therapeutic challenges due to neuronal loss, neuroinflammation, and complex genetics. The central nervous system’s intricate architecture, restrictive blood–brain barrier (BBB), and non-dividing neurons limit the effectiveness of traditional drugs.
Adeno-associated virus (AAV) vectors have emerged as powerful tools for central nervous system (CNS) gene delivery, offering safe, efficient, and long-term transgene expression in non-dividing neurons. Their low immunogenicity and high transduction fidelity make them ideal for neurological applications, accelerating the development of transformative AAV therapies.
The success of Zolgensma (AAV9–SMN1), the first FDA-approved AAV product for a neurological disease, validated systemic AAV delivery as a viable therapeutic strategy. This breakthrough has since accelerated the development of AAV-based treatments for diverse CNS disorders, driving significant growth in viral vector production and AAV manufacturing. This review summarizes current advances in AAV serotypes, AAV capsid engineering, and delivery strategies, along with emerging clinical applications.
AAV Applications in Neuroscience Research
AAV vectors have become indispensable tools in neuroscience research, enabling precise genetic manipulation of neural circuits and cells with high efficiency and minimal toxicity. Unlike conventional tracers, AAVs carry nucleic acid-encoded information that can be engineered for desired properties, such as expressing fluorescent reporters, actuators, sensors, or disease-related genes. This allows space-specific and time-specific interventions, revolutionizing studies of brain function, connectivity, and pathology.
Circuit Identification and Neuronal Tracing
AAV vectors excel in mapping neural connections by delivering fluorescent reporters to label cells and trace pathways. For instance, AAVs express proteins like GFP or mCherry to visualize neuronal morphology, axonal projections, and synaptic connections.
Circuit Manipulation
AAVs deliver actuators for precise control of neuronal activity, supporting functional studies. Optogenetics involves light-sensitive channels like Channelrhodopsin-2 (ChR2) for activation or halorhodopsin (NpHR) for inhibition. Chemogenetics uses designer receptors (DREADDs) activated by inert ligands.
Activity Monitoring
AAV vectors express genetically encoded sensors to monitor neural dynamics in vivo. Calcium indicators like GCaMP detect firing rates via fluorescence changes, while neurotransmitter sensors provide real-time readout of synaptic transmission.
Disease Modeling
AAVs model neurological disorders by expressing pathogenic proteins or suppressing genes, overcoming limitations of transgenic animals. AAV construction is key here, enabling gain-of-function models (e.g., Huntington’s, Parkinson’s, Alzheimer’s) or loss-of-function models (e.g., SMA, lysosomal storage diseases).

Figure 1. AAV applications in neuroscience research
AAV Serotypes and Vector Engineering
Natural AAVs are limited in efficiency and precision for CNS gene delivery, often requiring invasive methods. The successful AAV production of clinical-grade material requires robust upstream processes, starting with meticulous Plasmid Preparation and GMP plasmid manufacturing to generate the necessary GMP plasmids for high-quality AAV packaging. For complex projects, many researchers and developers rely on a dedicated plasmid service or plasmid CDMO for reliable plasmid production service.
The goal for vector engineering is to develop precisely targeted, minimally invasive recombinant vectors while maximizing the 4.7-kb cargo capacity. AAV advancement uses two strategies:
- AAV Capsid Engineering:
Capsid engineering further refines viral vector properties to enhance tropism and transduction efficiency. Techniques like site-directed mutagenesis and directed evolution have produced variants such as AAV-DJ for broad transduction, and BI-hTFR1, AAV-PHP.eB, and AAV-PHP.B for efficient CNS crossing via intravenous delivery.
For targeting challenging cells like microglia—critical in neuroinflammation—engineered capsids such as AAVTM6 and AAV-cMG achieve up to 86% transduction in primary cultures, though their in vivo efficiency remains modest (e.g., 3-18% in the spinal cord) due to biological barriers. Other innovative strategies, including mosaic or peptide-modified capsids and exosome-enveloped AAVs, achieve high CNS potency at dramatically lower doses. For instance, peptide fusions targeting the transferrin receptor (TfR) or PB5-3-modified AAV9 have yielded two-fold higher enzyme expression in mouse models of MPS I. Collectively, these engineering advances promise to expand the treatable disease spectrum while improving safety and manufacturability.
This process Creates recombinant/engineered capsids with unique tropisms (e.g., broad CNS/PNS transduction, cell type specificity, retrograde transport) for specific and efficient brain entry. This often involves running an AAV library screen (like PackGene π-Icosa™ AAV serotype screening platform) to identify tissue-specific and highly infective serotypes. This platform facilitates the development of custom AAV vectors. We also offer a dedicated AAV construction service to support the creation of novel vectors.

Figure 2. π-Icosa™ AAV serotype screening platform

Figure 3. AAV capsids for neuroscience
The AAV serotype is a foundational choice in CNS gene therapy, as each variant offers distinct advantages for specific applications. Below are the commonly used serotypes (Table 1):
- AAV2, the archetypal local delivery vector, provides high neuronal transduction at stereotactic injection sites and remains widely used in Parkinson’s and Alzheimer’s disease trials to deliver neurotrophic factors directly to structures like the striatum or hippocampus.
- AAV5 exhibits broader spread within the striatum, a property leveraged by uniQure’s AMT-130 to silence the mutant huntingtin gene.
- AAV9 represented a major advance by crossing the BBB after systemic administration, enabling motor neuron rescue in spinal muscular atrophy. However, high doses required revealed significant manufacturing and safety challenges.
- AAVhu.10 and AAVhu.68, derived from primates, display enhanced distribution through the cerebrospinal fluid and are being used in lysosomal storage disease trials to deliver therapeutic enzymes broadly across the CNS.

Table 1. AAV serotypes in neuroscience research and CNS gene therapy
- Genome Engineering:
This optimizes the recombinant AAV (rAAV) genome—specifically the ITR and promoter—by identifying short gene regulatory elements. This enables precise control of transgene expression level, cell-type specificity, and duration while staying within the vector’s size limit.
Cell-type-specific promoters and microRNA target sites can fine-tune expression and reduce off-target toxicity. Ubiquitous promoters (e.g., CMV, CBA) enable high-level expression, while neuron-specific (e.g., hSyn, CaMKII) and microglia-specific (e.g., F4/80, CD68) promoters enable targeted delivery. Taysha’s TSHA-102 for Rett syndrome exemplifies this approach with a self-regulating MECP2 cassette designed to prevent overexpression toxicity.
Incorporating microRNA target sequences (e.g., miR-9T) can detarget non-microglial cells, improving specificity as seen in strategies for neuroinflammation in Alzheimer’s and Parkinson’s disease. Finally, post-transcriptional elements like the WPRE enhance RNA stability, and inteins enable split-gene delivery for larger genetic payloads.
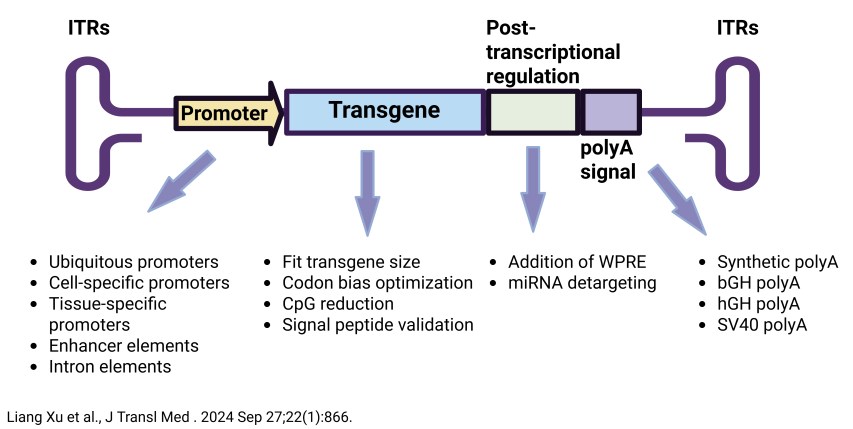
Figure 4. AAV genome cassette engineering strategy
AAV Delivery Routes and Cellular Targets in the CNS
AAV vectors can be delivered by several routes, each with specific advantages:
- Intraparenchymal (IP) delivery provides high local concentration for deep brain targeting. Intrastriatal (IStr) injections (e.g., in Huntington’s disease, AMT-130) transduce neurons in the caudate/putamen, while intrathalamic (ITh) injections reach cortical–subcortical relay hubs, relevant for epilepsy and circuit mapping.
- Intrathecal, intracisternal, or intracerebroventricular (IT/ICM/ICV) routes achieve broad CNS coverage with minimal systemic exposure, suitable for lysosomal and neurodevelopmental disorders. These routes also reach astrocytes and oligodendrocytes, critical for diseases like ALS and demyelination.
- Systemic intravenous (IV) delivery offers widespread transduction (e.g., Zolgensma for SMA) but is limited by hepatotoxicity, immune responses, and high cost. New capsids and promoters are improving microglial and CNS-wide targeting.

Figure 5. AAV Delivery Route to CNS

Table 2. AAV Delivery Routes to the Central Nervous System in Preclinical Disease Models
Disease-Specific Animal Models with AAV
AAV vectors enable precise modeling of neurological and psychiatric diseases by targeting specific brain regions and circuits. Below are examples of neuropsychiatric, neurodegenerative, and anatomical models that can be generated using AAV.
- Depression: SIRT1/CRTC1 modulation in amygdala or hippocampus.
- Movement & Epilepsy: GNAO1 mutations in striatum.
- Sleep: Orexin targeting in hypothalamus.
- Alzheimer’s: APP/tau/PSEN1 expression in hippocampus or cortex.
- Parkinson’s: α-Synuclein or LRRK2 in substantia nigra.
- ALS: TDP-43, SOD1, or C9orf72 models motor neuron loss.
- Huntington’s/Ataxia: Striatal or cerebellar targeting for neurodegeneration studies.
Key Regions: Amygdala, hippocampus, striatum, substantia nigra, hypothalamus.
Clinical Translation of AAV Gene Therapies
The AAV therapies pipeline for CNS disorders has grown rapidly. The transition from discovery to clinical reality requires scaled, compliant viral vector manufacturing and AAV preparation. Consequently, there is an urgent need for specialized providers offering comprehensive AAV packaging service, AAV production service, and AAV manufacturing service. To ensure the production of clinical-grade GMP AAV vectors suitable for trials, many companies partner with an AAV CDMO. PackGene is the trusted partner for this, ensuring strict regulatory adherence starting with GMP plasmid manufacturing which is critical for overall regulatory success.
Serotypes such as AAV9, AAVrh10, and engineered variants are favored for their BBB penetration and broad neuronal transduction via systemic or intrathecal delivery. Two CNS-targeted AAV therapies are approved—Zolgensma (AAV9-SMN1) for SMA and Kebilidi/Upstaza (AAV2-AADC) for AADC deficiency—while several others are in pivotal or registrational stages. Several are advancing toward late stages:
- uniQure – AMT-130 (Huntington’s): AAV5-miHTT via striatal infusion; ~75% slowing of progression over 3 years; FDA Breakthrough (2025); BLA in 2026.
- REGENXBIO – RGX-121 (Hunter syndrome): AAV9-IDS intrathecal; ~85% CSF HS reduction for 2 years; FDA Priority Review, PDUFA Nov 2025.
- Taysha – TSHA-102 (Rett): scAAV9-MECP2 intrathecal; FDA Breakthrough and positive regulatory feedback (Oct 2025).
- VectorY & Shape Tx – SHP-DB1 ($1.2B): Engineered AAV5 for IV antibody delivery to deep brain regions.

Table 3. The clinical status of AAV gene therapy for CNS disease
Conclusion
AAV gene therapy is entering an era of clinical reality for neurological disease. Advances in AAV capsid engineering, delivery methods, regulatory science, and strategic AAV CDMO partnerships are overcoming the historic barriers of the brain. With pivotal programs maturing and next-generation platforms emerging, the coming years are likely to bring the first AAV therapies approvals for adult neurodegenerative and neurodevelopmental disorders—while continuing to power discovery and disease modeling across neuroscience.
Author: Jin Qiu
References:
- Lunev E. et al. Adeno-Associated Viruses for Modeling Neurological Diseases in Animals: Achievements and Prospects. 2022; 10(5):1140.
- Castle M.J. et al. Controlling AAV Tropism in the Nervous System with Natural and Engineered Capsids. Methods Mol Biol. 2016; 1382:133–149.
- Dou D. et al. Key Challenges and Optimization Practices for Non-clinical Evaluation of AAV-based Gene Therapy for CNS Disease. Pharm Res. 2025. doi:10.1007/s11095-025-03931-2.
- Huang Q. et al. An AAV Capsid Reprogrammed to Bind Human Transferrin Receptor Mediates Brain-Wide Gene Delivery. 2024; 384(6701):1220–1227. doi:10.1126/science.adm8386.
- Zhou L. et al. A Comprehensive Review of AAV-Mediated Strategies Targeting Microglia for Neurodegenerative Diseases. J Neuroinflammation. 2024; 21:232.
- Haggerty D.L. et al. Adeno-Associated Viral Vectors in Neuroscience Research. Mol Ther Methods Clin Dev. 2019; 17:69–82.
- Challis R.C. et al. Adeno-Associated Virus Toolkit to Target Diverse Brain Cells. Annu Rev Neurosci. 2022; 45:447–469.
- Xu L. et al. Designing and Optimizing AAV-Mediated Gene Therapy for Neurodegenerative Diseases: From Bench to Bedside. J Transl Med. 2024; 22:866.
- Hudry E. et al. Therapeutic AAV Gene Transfer to the Nervous System: A Clinical Reality. 2019; 102:263.
- Minetti A. Unlocking the Potential of Adeno-Associated Virus in Neuroscience: A Brief Review. Mol Biol Rep. 2024; 51:563.
About PackGene
PackGene Biotech is a world-leading CRO and CDMO, excelling in AAV vectors, mRNA, plasmid DNA, and lentiviral vector solutions. Our comprehensive offerings span from vector design and construction to AAV, lentivirus, and mRNA services. With a sharp focus on early-stage drug discovery, preclinical development, and cell and gene therapy trials, we deliver cost-effective, dependable, and scalable production solutions. Leveraging our groundbreaking π-alpha 293 AAV high-yield platform, we amplify AAV production by up to 10-fold, yielding up to 1e+17vg per batch to meet diverse commercial and clinical project needs. Moreover, our tailored mRNA and LNP products and services cater to every stage of drug and vaccine development, from research to GMP production, providing a seamless, end-to-end solution.



